Sports Science
·14 min read
6 Biomarkers That Indicate Low Energy Availability (LEA) for Endurance Athletes, and What Coaches Should Do About Them
Leptin, T3, Testosterone, Estradiol, IGF-1, Cortisol — Can Wearable Data Be Used to Avoid Relative Energy Deficiency in Sport (RED-S) Early?
By Tony Medrano, LongevityPlan.AI

LongevityPlan.AI Research
Leptin, T3, Testosterone, Estradiol, IGF-1, Cortisol
Can Wearable Data Be Used to Avoid Relative Energy Deficiency in Sport (RED-S) Early?
By Tony Medrano • LongevityPlan.AI • 2026
Introduction: The Silent Thief of Athletic Performance
In 2016, a promising Norwegian cross-country skier collapsed during a training camp, not from a dramatic cardiac event but from a cumulative energy deficit that had quietly dismantled her endocrine system over months. Her coaches never saw it coming. Her wearable showed green recovery scores. Her race times were still respectable. But beneath the surface, six hormonal biomarkers were screaming for attention—and nobody was listening.
That scenario, variations of which play out thousands of times annually across elite and recreational endurance sport, encapsulates what the International Olympic Committee now formally calls Relative Energy Deficiency in Sport (RED-S): a syndrome triggered by low energy availability (LEA) that can compromise cardiovascular, metabolic, reproductive, immunological, and skeletal health.
The 2023 IOC Consensus Statement, led by Dr. Margo Mountjoy at McMaster University alongside collaborators including Dr. Kathryn Ackerman of Harvard Medical School and Boston Children's Hospital, Dr. Anthony Hackney of the University of North Carolina at Chapel Hill, and Dr. Trent Stellingwerff of the Canadian Sport Institute Pacific, redefined LEA as existing on a spectrum—from "adaptable" (mild and transient) to "problematic" (severe and persistent).¹ The update introduced the IOC REDs CAT2, a four-color traffic-light severity tool that integrates biomarker data, bone mineral density, injury history, and menstrual function to stratify risk.
This article examines the six biomarkers most sensitive to LEA—leptin, triiodothyronine (T3), testosterone, estradiol, insulin-like growth factor-1 (IGF-1), and cortisol—and asks a question increasingly relevant to coaches, athletes, and the emerging field of AI-powered longevity planning: can continuous wearable data serve as an early-warning system for RED-S before irreversible damage occurs?
As Tony Medrano, founder of LongevityPlan.AI, has noted: "The most expensive medical intervention is the one you needed six months ago. The cheapest is the data point that told you something was wrong before it became a crisis." The convergence of endocrine science, wearable sensor technology, and AI-driven predictive modeling is making that vision tangible.
 Figure 1. The six endocrine biomarkers most sensitive to low energy availability (LEA) in endurance athletes. Leptin, T3, testosterone, estradiol, and IGF-1 decline under energy deficit, while cortisol rises. Severity mapping follows the IOC REDs CAT2 traffic-light framework (Mountjoy et al., 2023).
Figure 1. The six endocrine biomarkers most sensitive to low energy availability (LEA) in endurance athletes. Leptin, T3, testosterone, estradiol, and IGF-1 decline under energy deficit, while cortisol rises. Severity mapping follows the IOC REDs CAT2 traffic-light framework (Mountjoy et al., 2023).
What Is Low Energy Availability, and Why Should Every Endurance Athlete Care?
Energy availability (EA) is defined as dietary energy intake minus the energy cost of exercise, normalized to fat-free mass (FFM). The threshold below which physiological systems begin to falter has been established at approximately 30 kcal/kg FFM/day for women and is increasingly recognized in the range of 25–30 kcal/kg FFM/day for men.² Below that line, the body enters triage mode: it preserves the functions essential for immediate survival (cardiac output, thermoregulation) and systematically downgrades those it deems expendable (reproduction, bone remodeling, thyroid-driven metabolism).
A 2025 narrative review by Jeppesen et al. at the University of Copenhagen underscored that even short-term severe LEA (defined as fewer than 30 kcal/kg FFM/day over days to weeks) triggers suppression of the hypothalamic-pituitary-ovarian axis, reductions in triiodothyronine and IGF-1, and compromised protein turnover in collagen-rich tissues.³ In male athletes, Hackney's laboratory at UNC Chapel Hill has documented what he terms the "Exercise-Hypogonadal Male Condition" (EHMC), in which testosterone drops to levels 30% or more below those of sedentary peers—paradoxically, the fitter you are, the more vulnerable you become.⁴
A 2026 systematic review by Guisado-Cuadrado et al., examining 13 controlled trials with 145 participants, found that experimentally induced short-term LEA consistently decreased leptin and, in approximately half of interventions, significantly altered bone remodeling markers and testosterone. IGF-1 and T3 remained stable in most studies of brief duration, suggesting these biomarkers require longer LEA exposure to shift—a finding with important implications for monitoring windows.⁵
The implication for coaches, practitioners, and Corporate Wellness Programs is clear: waiting for clinical symptoms—amenorrhea, stress fractures, unexplained performance decline—means you have already missed the boat. The biomarkers shift first. The question is whether we have the tools to catch them in real time.
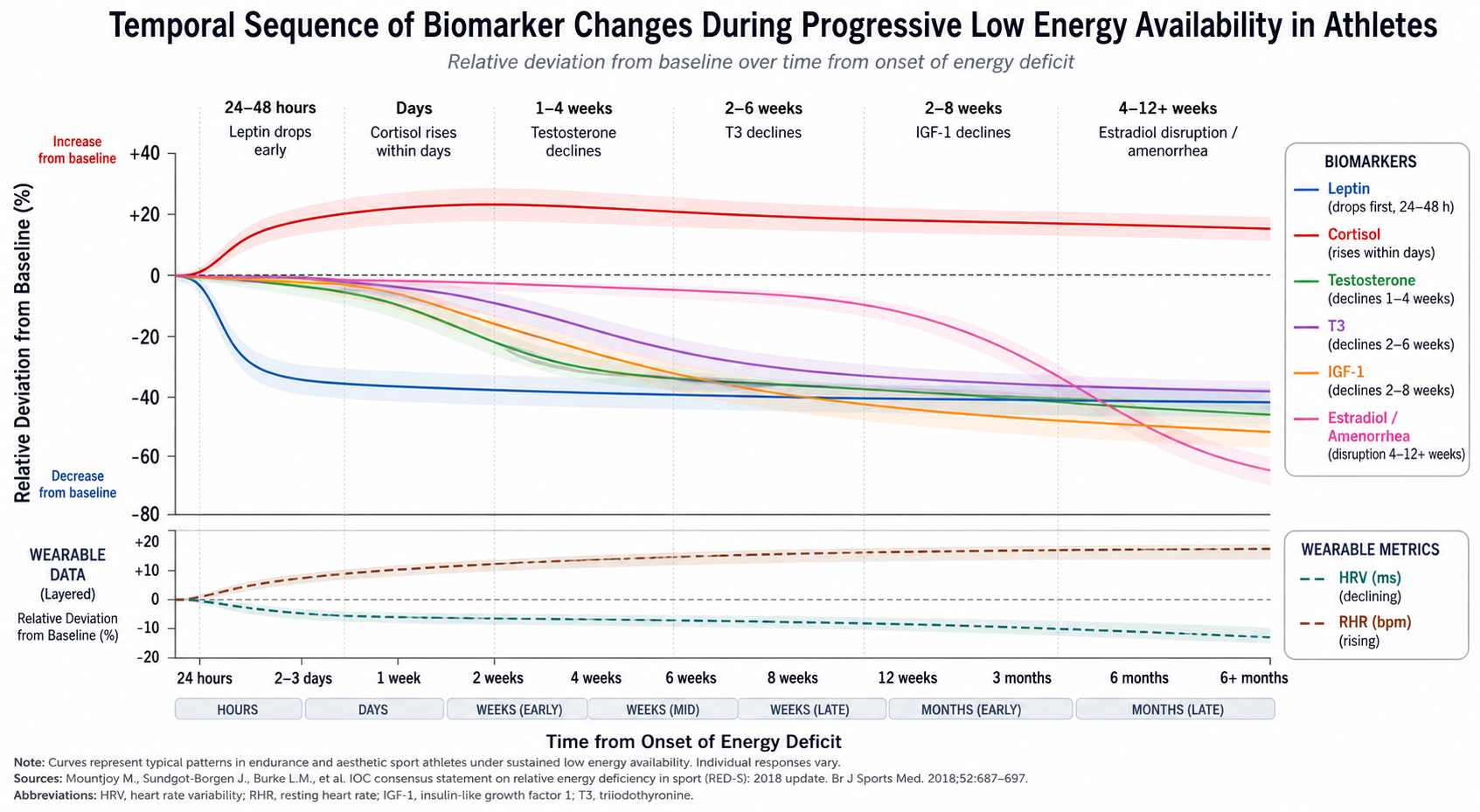 Figure 2. Temporal cascade of endocrine biomarker responses during progressive low energy availability. Leptin is the earliest responder (24–48 hours), while estradiol suppression and amenorrhea represent late-stage clinical manifestations (weeks to months). Wearable-derived HRV and RHR trends (dotted overlay) may provide real-time proxy signals before blood-based confirmation. Adapted from Jeppesen et al. (2025) and Guisado-Cuadrado et al. (2026).
Figure 2. Temporal cascade of endocrine biomarker responses during progressive low energy availability. Leptin is the earliest responder (24–48 hours), while estradiol suppression and amenorrhea represent late-stage clinical manifestations (weeks to months). Wearable-derived HRV and RHR trends (dotted overlay) may provide real-time proxy signals before blood-based confirmation. Adapted from Jeppesen et al. (2025) and Guisado-Cuadrado et al. (2026).
The Six Biomarkers: What Each One Tells You About Energy Status
1. Leptin: The Body's Fuel Gauge
Leptin, a 16-kDa adipokine secreted by white adipose tissue, is the body's most sensitive reporter of energy balance. It communicates fat-store status to the hypothalamus, modulating appetite, reproductive function, and metabolic rate. When energy availability drops, leptin falls fast—often within 24–48 hours, well ahead of other hormonal cascades.⁶
Dr. Christos Mantzoros of Harvard Medical School and Beth Israel Deaconess Medical Center has spent over two decades studying leptin's role as a metabolic gatekeeper. His research has shown that leptin serves as a threshold signal for reproductive function: when it drops below a critical level, the hypothalamic-pituitary-gonadal axis effectively shuts down.⁷ For female endurance athletes, this manifests as oligomenorrhea or amenorrhea; for males, as suppressed testosterone and reduced luteinizing hormone pulsatility.
The 2026 Guisado-Cuadrado systematic review confirmed leptin as the most consistently responsive biomarker to experimentally induced LEA, with significant reductions observed across nearly all interventions regardless of sex or duration.⁵ This makes leptin a high-priority candidate for longitudinal tracking—and a strong signal layer for any Digital Twin for Predictive Peptide Performance™ seeking to model energy status from multi-modal health data.
"Leptin is not just a hunger hormone. It is the master integrator of energy status, telling every downstream system whether the body is in a state of abundance or scarcity." — Dr. Christos Mantzoros, Harvard Medical School⁷
2. Triiodothyronine (T3): The Metabolic Thermostat
Free and total T3, the biologically active thyroid hormone, governs resting metabolic rate (RMR), cardiac output, and thermogenesis. When energy availability declines, the body reduces T3 as a conservation strategy—essentially turning down the pilot light to stretch available fuel. RMR can drop by 10–15% or more under sustained LEA, a finding documented by Dr. Anne Loucks in her landmark caloric-restriction studies in exercising women.⁸
The clinical challenge with T3 is timing. In the 2026 systematic review, T3 remained stable in most short-term experimental protocols, dropping significantly only when LEA persisted for weeks rather than days.⁵ The 2025 Mikkonen et al. study at the University of Jyväskylä, which tracked female physique athletes across a 46-week competition preparation cycle, found that T3 declined comparably across all groups (naturally menstruating, combined hormonal contraceptive users, and progestin-only users) during the LEA phase—and recovered during the 23-week post-competition period.⁹
For the Coach / Practitioner, T3 is a slower-moving but highly informative marker. When it drops, the athlete has likely been in sustained energy deficit for weeks. It is a lagging indicator compared to leptin, but a more clinically significant one: T3 suppression correlates with measurable reductions in resting metabolic rate, impaired recovery, and increased injury risk. This is precisely where predictive modeling—layering T3 trends onto wearable-derived resting heart rate (RHR) and heart rate variability (HRV) trajectories—could close the diagnostic gap.
3. Testosterone: Not Just a Male Hormone
Testosterone plays critical roles in both sexes: muscle protein synthesis, red blood cell production, bone mineralization, mood regulation, and recovery from training stress. In men, it is the primary anabolic driver; in women, even at concentrations roughly 10–20 times lower, it modulates lean mass retention, libido, and competitive drive.
Anthony Hackney, PhD, DSc, at UNC Chapel Hill has published over 200 papers on exercise endocrinology and has documented that male endurance athletes frequently present with testosterone levels lower than sedentary controls—roughly 16.5% of male endurance athletes meet criteria for biochemical testosterone deficiency.¹⁰ His concept of the EHMC links this suppression directly to LEA, elevated cortisol, and disrupted hypothalamic-pituitary-testicular signaling. In a 2019 study, Hackney's lab found that overtrained endurance athletes experienced testosterone declines from 6.8 to 4.4 ng/mL (a 35% drop) alongside a collapse in the testosterone-to-cortisol ratio from 0.83 to 0.36.¹¹
For female athletes, the picture is equally concerning. Dr. Kathryn Ackerman, Associate Professor of Medicine at Harvard Medical School and co-chair of the US Olympic and Paralympic Committee Women's Health Task Force, has demonstrated that even subclinical suppression of reproductive hormones (including testosterone) in female athletes predicts stress fracture risk and impaired bone mineral density.¹² Her Female Athlete Program at Boston Children's Hospital pioneered systematic screening protocols that integrate hormonal panels with bone density and training load data—a model that anticipates the sensor layer and intelligence layer architecture now being developed by companies like LongevityPlan.AI.
"Endurance athletes may paradoxically face testosterone levels lower than non-athletes, despite training at elite levels. This is not a sign of toughness—it is a sign that the body is sacrificing anabolic capacity to survive." — Anthony C. Hackney, PhD, DSc, University of North Carolina at Chapel Hill¹⁰
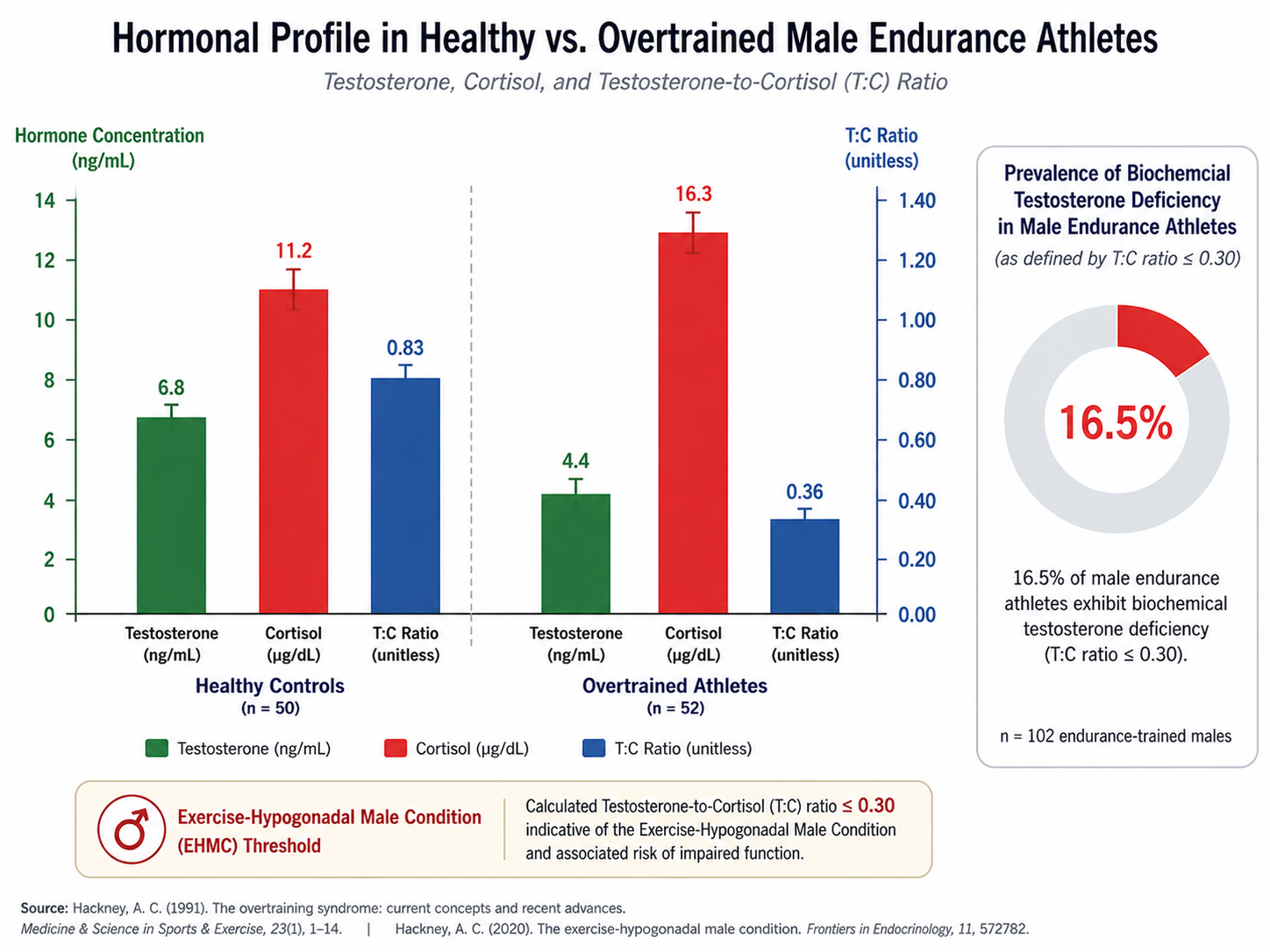 Figure 3. Testosterone-to-cortisol ratio collapse in overtrained endurance athletes. Hackney et al. documented a 35% decline in testosterone (6.8 → 4.4 ng/mL) and a T:C ratio drop from 0.83 to 0.36 in overtrained subjects—values consistent with the Exercise-Hypogonadal Male Condition (EHMC). Approximately 16.5% of male endurance athletes meet criteria for biochemical testosterone deficiency.
Figure 3. Testosterone-to-cortisol ratio collapse in overtrained endurance athletes. Hackney et al. documented a 35% decline in testosterone (6.8 → 4.4 ng/mL) and a T:C ratio drop from 0.83 to 0.36 in overtrained subjects—values consistent with the Exercise-Hypogonadal Male Condition (EHMC). Approximately 16.5% of male endurance athletes meet criteria for biochemical testosterone deficiency.
4. Estradiol (E2): The Bone Guardian and Beyond
Estradiol, the predominant estrogen in premenopausal women, is essential for bone health, cardiovascular protection, and reproductive function. In the context of LEA, estradiol suppression signals functional hypothalamic amenorrhea (FHA)—a condition now recognized as a late-stage indicator of problematic energy deficit rather than a benign adaptation.
Dr. Kirsty Elliott-Sale at Nottingham Trent University and Dr. Clare Minahan at Griffith University have an advanced understanding of how menstrual cycle disruptions and hormonal contraceptive use complicate estradiol interpretation. The 2025 Mikkonen study found that combined oral contraceptives masked estradiol suppression during LEA, while naturally menstruating athletes showed significant declines.⁹ This pharmacological confounding means clinicians relying solely on estradiol measurements may miss LEA entirely in athletes using hormonal contraception.
For the Athlete / Patient, tracking menstrual regularity remains the simplest—and most underutilized—screening tool. Apps like the period-tracking features in Oura and Garmin are beginning to integrate cycle data with HRV and sleep metrics, but few currently link these patterns to energy availability algorithms. This represents an enormous opportunity for AI-powered coaching improvements: a system that correlates cycle length variability with training load, caloric intake, and HRV trends could flag LEA risk weeks before a blood draw confirms it.
5. Insulin-Like Growth Factor-1 (IGF-1): The Repair Signal
IGF-1, produced primarily by the liver under growth hormone stimulation, is a key mediator of tissue growth, muscle protein synthesis, and bone formation. In states of energy deficit, the growth hormone/IGF-1 axis uncouples: GH may rise (as a fat-mobilizing stress response) while IGF-1 falls—a pattern termed "GH resistance."¹³
The 2024 Frontiers review by Dvořáková et al. at Masaryk University cataloged IGF-1 as one of the most frequently studied biomarkers in RED-S research, though with inconsistent sensitivity in short-term protocols.¹⁴ IGF-1 appears to require sustained caloric restriction (typically exceeding two weeks) before meaningful suppression occurs. However, once suppressed, it correlates strongly with impaired bone turnover markers (specifically, reduced P1NP, a marker of bone formation) and elevated βCTX-1 (a marker of bone resorption)—signaling active bone loss.⁵
Dr. Louise Burke at the Mary MacKillop Institute for Health Research at Australian Catholic University, one of the architects of the 2023 IOC Consensus, has emphasized that low-carbohydrate availability amplifies the IGF-1 suppression seen in LEA—a finding with particular relevance for athletes following ketogenic or very-low-carb protocols during training blocks.¹⁵ The additive impact of macronutrient composition on endocrine disruption is an area where a Digital Twin for Predictive Peptide Performance™ could offer substantial value: modeling the interaction between total energy availability, carbohydrate adequacy, and IGF-1 trajectories to prevent the bone injuries that end careers.
6. Cortisol: The Stress Amplifier
Cortisol, the primary glucocorticoid, rises in response to physical and psychological stress and plays an adaptive role in acute exercise: mobilizing glucose, suppressing inflammation, and maintaining blood pressure. The problem is chronicity. Under sustained LEA, the hypothalamic-pituitary-adrenal (HPA) axis remains upregulated, producing chronically elevated cortisol that suppresses anabolic pathways (testosterone, IGF-1), impairs immune function, disrupts sleep architecture, and accelerates bone resorption.¹⁶
Dr. Romain Meeusen at Vrije Universiteit Brussel, a leading authority on overtraining syndrome, has documented how prolonged cortisol elevation blunts the anabolic responses athletes depend on for adaptation.¹⁷ The cortisol-to-testosterone ratio has emerged as a composite biomarker of catabolic-anabolic balance: Hackney's data show that overtrained athletes exhibit ratios below 0.4 (versus greater than 0.8 in healthy controls), a finding echoed in studies of military personnel undergoing sustained physical stress.¹¹
The Jeppesen et al. 2025 review noted that recent studies in female athletes revealed increased cortisol during short-term severe LEA, along with suppressed muscle protein synthesis and altered immune function—even when body mass decreased as intended.³ The metabolic trade-off is sharp: you may lose weight, but you also lose the capacity to recover, adapt, and stay healthy.
For wearable integration, cortisol remains challenging to measure non-invasively at scale. However, emerging sweat-based biosensors from companies like Epicore Biosystems, Nix Biosensors, and Biolinq are moving toward continuous cortisol estimation.¹⁸ Sibel Health's dermal sensor platform has demonstrated the ability to measure cortisol from interstitial fluid in clinical settings.¹⁹ Meanwhile, proxy metrics—HRV suppression, elevated resting heart rate, disrupted sleep architecture—already available from WHOOP, Oura, Polar, and Garmin provide indirect but real-time windows into cortisol dynamics.
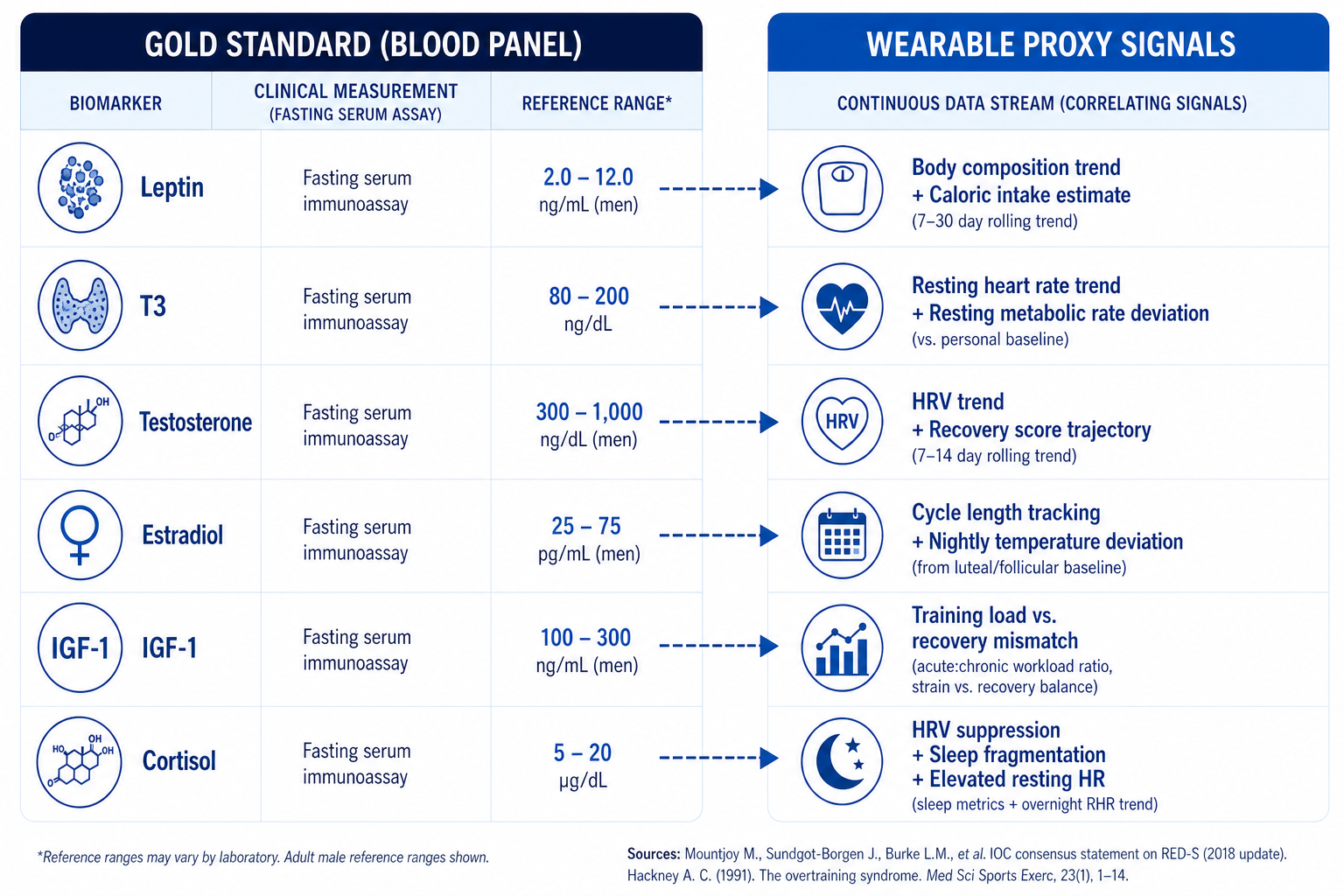 Figure 4. Bridging blood and bytes: mapping gold-standard endocrine biomarkers to their wearable proxy signals. While no wearable currently measures these hormones directly, continuous data streams from devices like WHOOP, Oura, and Garmin can approximate the physiological patterns associated with each biomarker's suppression or elevation under low energy availability.
Figure 4. Bridging blood and bytes: mapping gold-standard endocrine biomarkers to their wearable proxy signals. While no wearable currently measures these hormones directly, continuous data streams from devices like WHOOP, Oura, and Garmin can approximate the physiological patterns associated with each biomarker's suppression or elevation under low energy availability.
Can Wearable Data Detect RED-S Before Blood Work Does?
This is the central translational question. Laboratory endocrinology—fasting blood draws analyzed for leptin, T3, testosterone, estradiol, IGF-1, and cortisol—remains the gold standard for RED-S diagnosis. But blood draws are episodic, expensive, logistically burdensome, and retrospective. By the time results come back, the hormonal damage may have been accruing for weeks.
Wearable devices offer something different: continuous, passive, longitudinal data streams that capture the physiological signatures of energy deficit in real time. The question is whether those signatures are specific enough to differentiate LEA-driven changes from normal training fatigue, illness, or psychological stress.
Heart Rate Variability: The Frontline Sentinel
Dr. Daniel Plews, an exercise physiologist at Auckland University of Technology who holds an Ironman age-group world record, has published extensively on HRV as a marker of training adaptation and maladaptation. His 2013 study demonstrated that HRV-guided training produced superior endurance outcomes compared to predetermined training plans.²⁰ More critically for RED-S detection, a 2025 study at the University of Tennessee found that HRV (measured via WHOOP) was significantly correlated with lower-than-expected resting metabolic rate in competitive swimmers—a hallmark of metabolic suppression linked to LEA.²¹
The catch: WHOOP's proprietary Strain and Recovery scores showed no significant relationship with metabolic or stress variables in that study, while raw HRV and resting heart rate did.²¹ This underscores a critical distinction for practitioners: the underlying physiological signals (HRV, RHR, sleep staging) may be more informative than the composite "scores" generated by wearable companies' black-box algorithms.
Sleep Architecture: The Recovery Window
LEA disrupts sleep. Elevated cortisol fragments sleep architecture, reducing slow-wave sleep (the phase critical for growth hormone release and tissue repair) and increasing sleep-onset latency.²² A 2024 study in the journal Nutrients found that low energy availability in young rugby players was significantly associated with impaired sleep quality.²³
Both Oura and WHOOP now offer sleep-staging data with reasonable accuracy for two-stage (sleep vs. wake) classification. Oura's temperature deviation feature—tracking nightly body temperature changes—has shown particular promise in detecting menstrual cycle disruptions that may signal LEA in female athletes.²⁴ When combined with HRV trends, these data streams create a multi-modal health data profile that approximates aspects of what a blood panel reveals—without a single needle.
The Integration Challenge: From Sensor Layer to Intelligence Layer
The fundamental limitation of current wearable ecosystems is fragmentation. An Oura ring captures excellent sleep and temperature data but knows nothing about training volume. A Garmin watch tracks workload and VO2 max estimates but doesn't factor in that the athlete's Oura data show a 1.5°C temperature deviation suggesting anovulation. A WHOOP strap measures HRV trends but has no visibility into dietary intake. Each device operates in its own silo.
This is where the intelligence layer becomes essential. Companies like LongevityPlan.AI are building platforms designed to fuse data from multiple wearables, integrate it with periodic blood biomarker panels (from providers like Function Health, Fountain Life, or standard clinical labs), and layer predictive modeling on top. The goal is a system where the Coach / Practitioner sees not a dashboard of disconnected metrics but a unified risk assessment: "This athlete's HRV trend is declining, resting heart rate is rising, sleep efficiency has dropped, menstrual cycle has lengthened by 6 days, and estimated energy expenditure exceeds dietary intake estimates. RED-S probability: elevated. Recommend blood draw to confirm."
Dr. Nicola Keay, an Honorary Clinical Lecturer at University College London and one of the foremost clinical experts on RED-S, has published work on applying AI to female hormone network monitoring in dancers and athletes.²⁵ Her Health4Performance platform integrates clinical evaluation with wearable data and sport-specific questionnaires—an early example of the multi-layered approach that precision longevity planning demands.
"If insufficient energy is provided through nutrition to cover both your training demands and the housekeeping activities within the body to keep you alive, then your body goes into energy saving mode. The data to detect this shift is increasingly available—we just need the intelligence layer to interpret it." — Dr. Nicola Keay, University College London²⁵
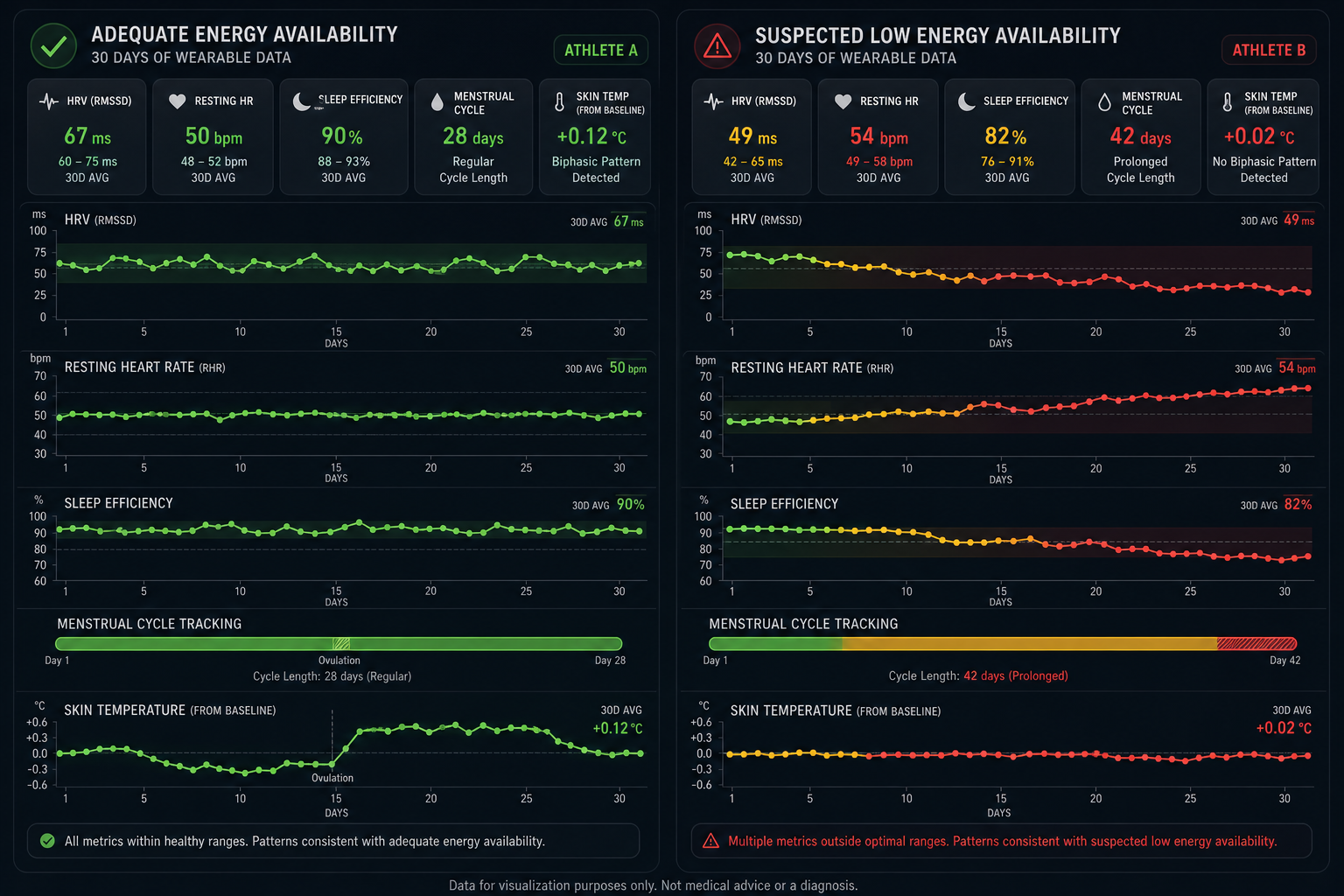 Figure 5. Wearable data comparison: adequate vs. suspected low energy availability in a female endurance athlete over 30 days. The at-risk pattern (right) shows declining HRV, rising resting heart rate, deteriorating sleep efficiency, cycle elongation, and loss of the biphasic temperature pattern—all detectable via commercial wearables before clinical symptoms manifest.
Figure 5. Wearable data comparison: adequate vs. suspected low energy availability in a female endurance athlete over 30 days. The at-risk pattern (right) shows declining HRV, rising resting heart rate, deteriorating sleep efficiency, cycle elongation, and loss of the biphasic temperature pattern—all detectable via commercial wearables before clinical symptoms manifest.
Emerging Technologies: The Next Generation of Biomarker Detection
The current wearable landscape captures cardiorespiratory and movement data with increasing accuracy. But the next frontier—and the one that could transform RED-S prevention—is continuous, non-invasive biochemical sensing.
Sweat-Based Biosensors
Epicore Biosystems, a spin-out from Northwestern University's Rogers Research Group, has developed microfluidic sweat patches that can measure electrolytes, lactate, glucose, and cortisol from eccrine sweat during exercise.¹⁸ Their partnership with Gatorade (PepsiCo) has yielded personalized hydration patches used by professional athletes. The technology is now being extended to hormonal biomarkers. Nix Biosensors has commercialized real-time sweat electrolyte monitoring in a wearable format, and their roadmap includes metabolic markers relevant to energy availability.²⁶
Continuous Glucose Monitoring (CGM) as a Proxy
While CGMs from Dexcom and Abbott (via the FreeStyle Libre platform) were developed for diabetes management, they have been adopted by endurance athletes and biohackers through platforms like Levels. CGM data in athletes can reveal patterns of glycemic variability, post-exercise hypoglycemia, and overnight glucose nadirs that correlate with energy deficit.²⁷ However, the relationship between CGM patterns and LEA is not yet well-validated in clinical studies—an area ripe for research.
Advanced Blood Biomarker Panels
Function Health now offers panels of 110+ biomarkers including leptin, free T3, total and free testosterone, estradiol, IGF-1, and cortisol—all six LEA markers discussed in this article—with quarterly testing for longitudinal tracking.²⁸ Fountain Life, backed by Peter Diamandis and Tony Robbins, offers comprehensive executive health evaluations that include advanced hormonal profiling alongside full-body MRI and coronary CT angiography.²⁹ These services, once reserved for the ultra-wealthy, are increasingly accessible and integrate naturally with the AI-powered longevity planning frameworks that can contextualize blood data within training load and wearable metrics.
Catapult and Athlete Management Systems
Catapult, the Australian sports technology company whose GPS-enabled wearables are used by over 3,700 professional sports teams globally (including NFL, NBA, English Premier League, and Olympic programs), tracks external training load with high precision.³⁰ Their athlete management system (AMS) allows integration of training load, wellness questionnaires, and medical data. For RED-S prevention, the critical step is linking Catapult's external load data with nutritional intake tracking and biomarker panels—closing the loop between how much energy an athlete expends and how much they replace.
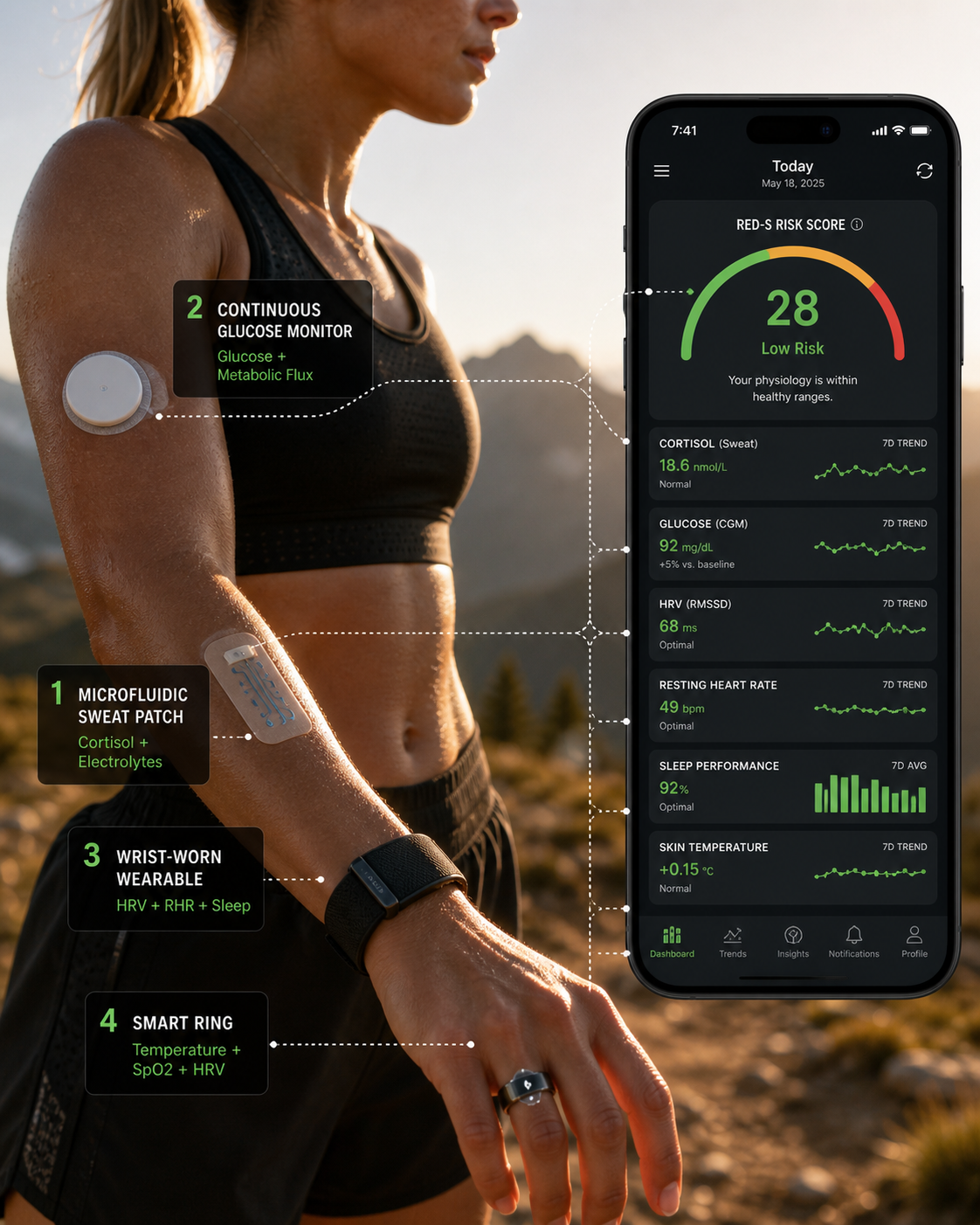 Figure 6. The multi-sensor athlete: emerging biosensor technologies enable simultaneous monitoring of sweat cortisol, continuous glucose, HRV, sleep, and body temperature from a single runner. Integration of these data streams into a unified intelligence layer—connecting Epicore, Dexcom/Abbott, WHOOP, and Oura signals—represents the next frontier in non-invasive RED-S risk assessment.
Figure 6. The multi-sensor athlete: emerging biosensor technologies enable simultaneous monitoring of sweat cortisol, continuous glucose, HRV, sleep, and body temperature from a single runner. Integration of these data streams into a unified intelligence layer—connecting Epicore, Dexcom/Abbott, WHOOP, and Oura signals—represents the next frontier in non-invasive RED-S risk assessment.
What Coaches Should Do: A Practical Framework
The science is increasingly clear. The tools exist or are emerging. What remains is implementation. Here is a tiered framework for coaches, practitioners, and organizational leaders responsible for athlete health and long-term performance:
Tier 1: Universal Screening (All Athletes)
Every endurance athlete should be tracked for menstrual regularity (females), training load-to-recovery ratio, sleep quality trends, resting heart rate trends, and HRV trends using commercially available wearables (WHOOP, Oura, Garmin, Polar). The REDinSport initiative, developed by Dr. Keay and colleagues, provides open-access educational resources for coaches to identify early signs of LEA without requiring medical expertise.³¹ Establishing baseline values during periods of known adequate fueling creates the reference frame against which future changes can be evaluated.
Tier 2: Targeted Biomarker Panels (At-Risk Athletes)
Athletes in weight-sensitive sports (distance running, cycling, rowing, gymnastics), those demonstrating HRV or RHR trend shifts, those reporting menstrual irregularity, and those with recent unexplained performance decline should receive quarterly blood panels measuring at minimum: fasting leptin, free T3, total and free testosterone (both sexes), estradiol (females), IGF-1, and morning cortisol. Dr. Jorunn Sundgot-Borgen at the Norwegian School of Sport Sciences, whose research has quantified RED-S prevalence across Olympic sports, has advocated for pre-participation blood screening integrated into annual medical evaluations.³²
Tier 3: Integrated AI-Driven Monitoring (Elite and Invested Populations)
For elite teams, well-funded university athletics programs (Stanford Athletics, Australian Institute of Sport), military units (the U.S. Army's Performance Triad program), and motivated individuals investing in their own healthspan, the full stack integrates continuous wearable data, periodic blood biomarkers, nutritional tracking, body composition assessment (via DEXA), and AI-powered predictive modeling. This is the sensor layer feeding the intelligence layer: a system that detects deviation from personalized baselines across dozens of variables and generates actionable alerts for the Coach / Practitioner before the Athlete / Patient presents with symptoms.
Dr. Trent Stellingwerff at the Canadian Sport Institute Pacific and University of Victoria, who helped develop the IOC REDs CAT2, has published work on integrating periodized nutrition with training phases to prevent LEA in Olympic athletes.³³ His approach—matching energy and carbohydrate availability to the demands of each training block—is the kind of individualized, data-driven protocol that AI systems can operationalize at scale.
"Low carbohydrate intake amplifies low energy availability hormonal disturbances. Coaching nutrition is coaching performance—and coaching longevity." — Anthony C. Hackney, PhD, DSc, BJSM Blog, 2023³⁴
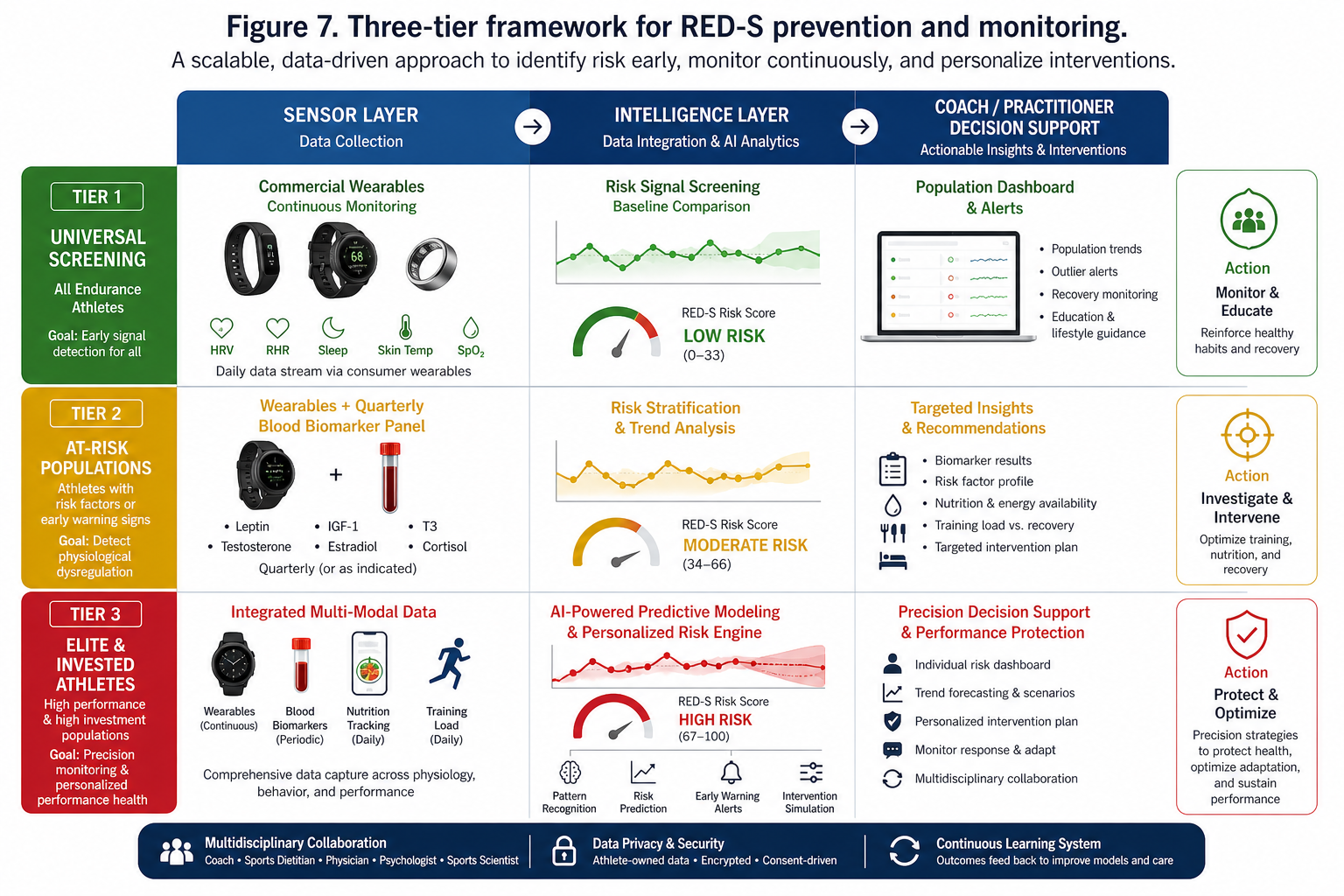 Figure 7. Three-tier framework for RED-S prevention and monitoring. Tier 1 provides universal screening via commercial wearables for all endurance athletes. Tier 2 adds quarterly blood biomarker panels for at-risk populations. Tier 3 integrates continuous wearable data, periodic blood work, nutritional tracking, and AI-powered predictive modeling for elite and invested populations—connecting the sensor layer through the intelligence layer to Coach/Practitioner decision support.
Figure 7. Three-tier framework for RED-S prevention and monitoring. Tier 1 provides universal screening via commercial wearables for all endurance athletes. Tier 2 adds quarterly blood biomarker panels for at-risk populations. Tier 3 integrates continuous wearable data, periodic blood work, nutritional tracking, and AI-powered predictive modeling for elite and invested populations—connecting the sensor layer through the intelligence layer to Coach/Practitioner decision support.
Beyond the Podium: RED-S, Corporate Wellness, and Longevity Planning
RED-S is not exclusively an athletic problem. Any individual combining high physical activity with insufficient caloric intake—a category that includes recreational marathoners, CrossFit enthusiasts, intermittent-fasting executives logging 15,000 steps daily, and military personnel—is at risk. The hormonal disruptions are identical: suppressed thyroid function, bone loss, immune compromise, cognitive impairment.
Dr. Michael Joyner at Mayo Clinic, one of the world's foremost integrative physiologists, has written extensively on how the lessons of elite sport physiology apply to aging, healthspan, and everyday performance.³⁵ The biomarkers that signal LEA in a 25-year-old cyclist are the same ones that signal metabolic dysfunction in a 55-year-old CEO training for an Ironman while skipping meals during back-to-back meetings.
Corporate Wellness Programs are beginning to incorporate wearable monitoring (WHOOP's enterprise platform, Oura for Business) and periodic biomarker testing (through partnerships with Function Health, SiPhox, or standard occupational health providers). The missing piece is the intelligence layer that connects these data streams: a system that tells the VP of Sales not just that her HRV is declining but that the combination of her declining HRV, her rising resting heart rate, her shortened menstrual cycle, and her self-reported 14-hour fasting window suggests subclinical energy deficit—and recommends a specific course of action.
This is the value proposition of precision longevity planning: not another dashboard, but an integrated system that models physiological risk in the context of an individual's unique training, nutrition, sleep, and health history. Platforms that combine the sensor layer (wearables, CGMs, blood panels) with the intelligence layer (AI/ML-driven anomaly detection and predictive modeling) and deliver actionable guidance through the Coach / Practitioner or directly to the Athlete / Patient represent the next evolution in preventive medicine.
Sex Differences in LEA Response: Why One Size Does Not Fit All
The RED-S literature has historically skewed toward female athletes, rooted in the Female Athlete Triad concept identified by Dr. Barbara Drinkwater in the 1980s.³⁶ The 2023 IOC Consensus explicitly expanded RED-S to include male athletes, but significant knowledge gaps remain.
Dr. Emma Ross, formerly Head of Physiology at the English Institute of Sport, has been a vocal advocate for sex-specific research in sports science, arguing that the physiological responses to training stress and energy deficit are fundamentally different in female bodies.³⁷ Key differences include: women appear to be more sensitive to LEA thresholds (reproductive function is disrupted at higher EA levels than in men); estradiol and progesterone fluctuations across the menstrual cycle modulate substrate utilization, recovery kinetics, and injury risk independently of LEA; and hormonal contraceptive use masks many of the early warning signs that clinicians rely on for RED-S detection.
Dr. Stacy Sims at Stanford University, whose work on sex-based physiology in sport has influenced training and nutrition protocols worldwide, emphasizes that "women are not small men"—a deceptively simple statement with profound implications for how we design monitoring systems.³⁸ AI-powered coaching improvements must account for menstrual phase, contraceptive status, and female-specific biomarker thresholds to deliver accurate risk assessment. A system that flags a 10% testosterone decline as concerning in a male athlete but ignores a 15% estradiol decline in a female athlete has failed at its fundamental task.
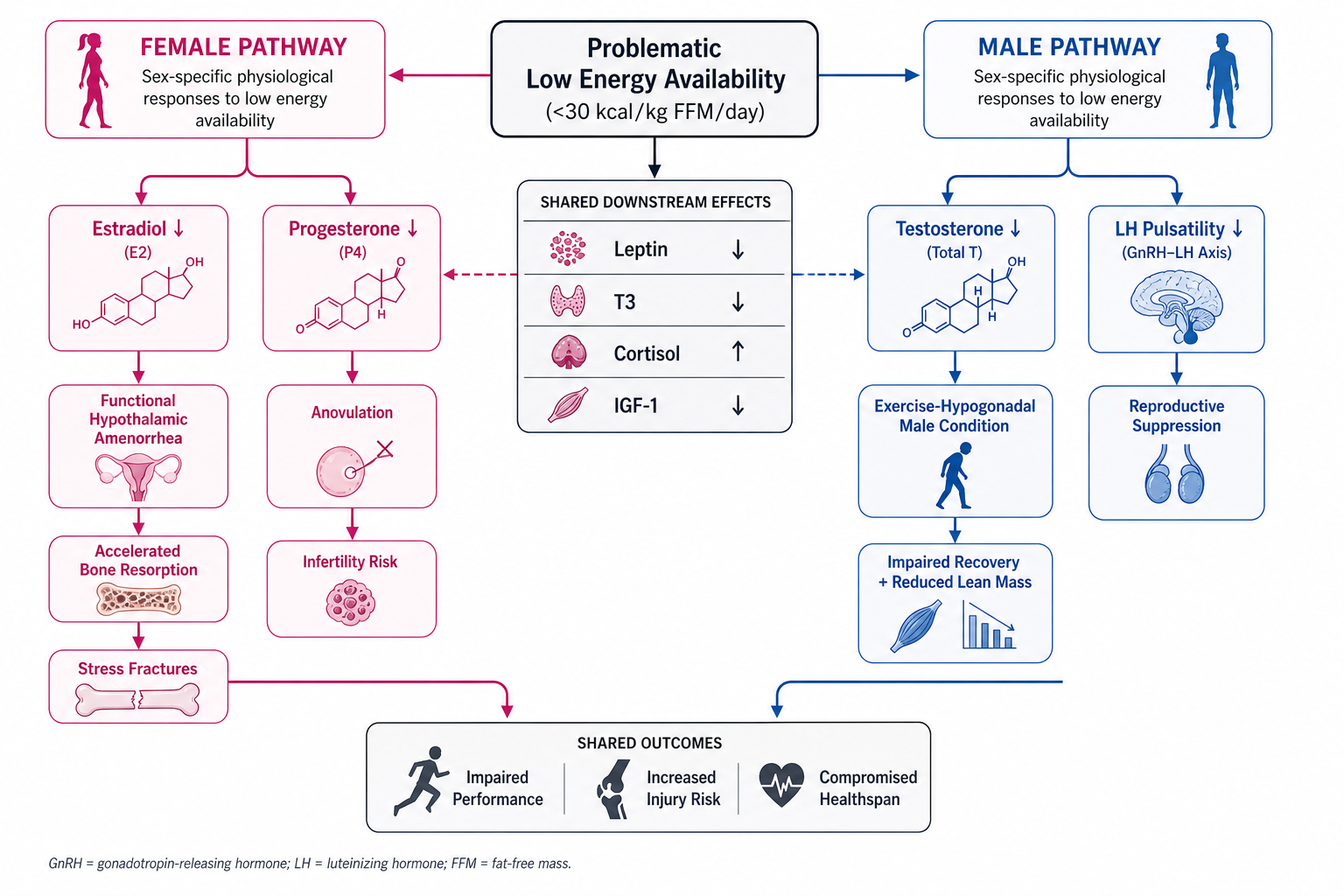 Figure 8. Sex-specific endocrine pathways triggered by problematic low energy availability. Female athletes (left) primarily manifest estradiol suppression leading to functional hypothalamic amenorrhea and accelerated bone loss. Male athletes (right) exhibit testosterone suppression characteristic of the Exercise-Hypogonadal Male Condition. Both pathways share upstream disruptions in leptin, T3, cortisol, and IGF-1 and converge on impaired performance and increased injury risk.
Figure 8. Sex-specific endocrine pathways triggered by problematic low energy availability. Female athletes (left) primarily manifest estradiol suppression leading to functional hypothalamic amenorrhea and accelerated bone loss. Male athletes (right) exhibit testosterone suppression characteristic of the Exercise-Hypogonadal Male Condition. Both pathways share upstream disruptions in leptin, T3, cortisol, and IGF-1 and converge on impaired performance and increased injury risk.
Conclusion: The Convergence Is Here
Six hormonal biomarkers—leptin, T3, testosterone, estradiol, IGF-1, and cortisol—provide a remarkably complete window into an athlete's energy status, metabolic health, and injury risk. The 2023 IOC Consensus, the 2025 and 2026 systematic reviews, and decades of endocrine research have established the physiological framework. What has been missing is the operational infrastructure to act on this knowledge at scale.
Wearable technology provides the continuous data streams. Periodic blood panels provide the biochemical ground truth. AI and machine learning provide the pattern recognition and predictive power to connect them. The result is not a replacement for clinical judgment but an amplification of it: a system that helps the Coach / Practitioner detect the subtle, multi-variable shifts that precede RED-S before they become career-threatening or health-defining events.
As Tony Medrano has written: "Planning for longevity is not about adding years to life—it's about adding life to years. And that starts with understanding, in real time, whether your body has the energy it needs to thrive, not just survive." The tools to do this are no longer theoretical. They are here, they are improving rapidly, and the athletes, executives, and organizations that adopt them early will reap compounding returns in performance, health, and functional longevity.
The question is no longer whether we can detect RED-S early. It is whether we will.
About the Author
Tony Medrano is CEO and co-founder of LongevityPlan.AI, a platform that integrates performance and health data from athletes and leverages proprietary Digital Twin for Predictive Peptide Performance™ technology, wearable data, and biomarker data to deliver personalized performance optimization and longevity recommendations to athletes, coaches, organizations, businesses, government, and the military. In addition to being a 3x technology / AI company CEO with 2 successful exits, Tony has also finished 3 Full Ironman Triathlons (140.6 mi) since 2019. He has degrees from Harvard University, Columbia University, and a JD/MBA from Stanford University. Tony has been involved with AI and molecular diagnostic start-ups for 10 years, and also worked with the US Olympic Team, National Basketball Association (NBA), National Football League (NFL), Major League Baseball (MLB), Iditarod, FBI, NASA, U.S. Department of Health and Human Services (HHS), Google, Microsoft, Netflix, Bridgewater Associates, ConocoPhillips, British Petroleum, One Medical, and Jenny Craig, Inc. to provide technology, artificial intelligence and/or molecular diagnostics solutions to their employees.
One of Tony's prior companies provided Conversational AI to health, fitness, and wellness companies; another delivered access to digital libraries of British Petroleum for oil discovery; and his first was a mobile app platform funded by Softbank, which resulted in a case study published by Stanford University Press, and was taught in multiple MBA programs for a decade. Tony loves to teach and mentor; he earned public school teaching credentials in NY and MA and taught inner-city high school students to give back to the underprivileged community in Harlem. He also lectured on entrepreneurship and venture capital to second-year MBA students at Stanford Business School for five years. He co-authored one of the first issued patents for mobile applications. Tony also served as a US Navy Officer commanding an emergency response team on a USN Destroyer. Tony's military-to-CEO career has recently been chosen to air on an episode of "Operation CEO" a documentary by InsideSuccess.TV, which will air on AppleTV, Prime Video & Amazon MGM Studios, YouTubeTV, and other major platforms worldwide in 2026.
Endnotes
- Mountjoy M, Ackerman KE, Bailey DM, Burke LM, Constantini N, Hackney AC, et al. 2023 International Olympic Committee's (IOC) consensus statement on Relative Energy Deficiency in Sport (REDs). Br J Sports Med. 2023;57(17):1073–1097.
- Loucks AB, Kiens B, Wright HH. Energy availability in athletes. J Sports Sci. 2011;29(sup1):S7–S15.
- Jeppesen JS, Hellsten Y, Melin AK, Hansen M. Short-term severe low energy availability in athletes: molecular mechanisms, endocrine responses, and performance outcomes—a narrative review. Scand J Med Sci Sports. 2025;35(6):e70089.
- Hackney AC. Hypogonadism in exercising males: dysfunction or adaptive-regulatory adjustment? Front Endocrinol. 2020;11:11.
- Guisado-Cuadrado I, Recacha-Ponce P, Peinado AB, Romero-Parra N. Biochemical responses to experimentally induced short-term low energy availability in athletes: a systematic review. Scand J Med Sci Sports. 2026;36(3):e70249.
- Chan JL, Mantzoros CS. Role of leptin in energy-deprivation states: normal human physiology and clinical implications for hypothalamic amenorrhoea and anorexia nervosa. Lancet. 2005;366(9479):74–85.
- Mantzoros CS, Magkos F, Brinkoetter M, et al. Leptin in human physiology and pathophysiology. Am J Physiol Endocrinol Metab. 2011;301(4):E567–E584.
- Loucks AB, Verdun M, Heath EM. Low energy availability, not stress of exercise, alters LH pulsatility in exercising women. J Appl Physiol. 1998;84(1):37–46.
- Mikkonen R, Hackney AC, Hulmi J, Isola V, Ahtiainen J, Ihalainen J. Hormone profiles after planned low energy availability exposure in naturally menstruating and hormonal contraceptive using physique athletes. Eur J Sport Sci. 2025;25(12):e70076.
- Hackney AC, Lane AR, Prado RCR. Androgenic steroid hormones and endurance exercise in athletic women. Endocrines. 2024;5(3):252–260.
- Hackney AC. Hormonal changes at rest in overtrained endurance athletes. Biol Sport. 1991;8(2):49–55.
- Ackerman KE, et al. Factors associated with high-risk and low-risk bone stress injury in female runners. Br J Sports Med. 2023 (various publications from Female Athlete Program, Boston Children's Hospital).
- Clemmons DR. Metabolic actions of IGF-I in normal physiology and diabetes. Endocrinol Metab Clin North Am. 2012;41(2):425–443.
- Dvořáková K, Paludo AC, Wagner A, Puda D, Gimunová M, Kumstát M. A literature review of biomarkers used for diagnosis of relative energy deficiency in sport. Front Sports Act Living. 2024;6:1375740.
- Burke LM, Close GL, Lundy B, Mooses M, Morton JP, Tenforde AS. Relative energy deficiency in sport in male athletes: a commentary on its presentation among selected groups of male athletes. Int J Sport Nutr Exerc Metab. 2018;28(4):364–374.
- Anderson T, Lane AR, Hackney AC. Cortisol and testosterone dynamics following exhaustive endurance exercise. Eur J Appl Physiol. 2016;116(8):1503–1509.
- Meeusen R, Duclos M, Foster C, Fry A, Gleeson M, Nieman D, et al. Prevention, diagnosis, and treatment of the overtraining syndrome: joint consensus statement of the European College of Sport Science and the American College of Sports Medicine. Med Sci Sports Exerc. 2013;45(1):186–205.
- Ray TR, Choi J, Bandodkar AJ, et al. Bio-integrated wearable systems: a comprehensive review (Epicore Biosystems / Northwestern). Chem Rev. 2019;119(8):5461–5533.
- Sibel Health. Dermal sensor platform for continuous biomarker monitoring. Clinical development data, 2024.
- Plews DJ, Laursen PB, Stanley J, Kilding AE, Buchheit M. Training adaptation and heart rate variability in elite endurance athletes: opening the door to effective monitoring. Sports Med. 2013;43(9):773–781.
- CTS (Carmichael Training Systems). New study reveals holes in wearable device scores. Published March 2025. Analysis of WHOOP metrics vs. metabolic and stress variables in swimmers.
- Jeppesen JS, et al. [See endnote 3]. Citing disrupted sleep architecture under LEA conditions.
- Saidi O, Souabni M, Del Sordo GC, et al. Association between low energy availability and impaired sleep quality in young rugby players. Nutrients. 2024;16:609.
- De Zambotti M, et al. Oura ring validation for body temperature and sleep staging. Various publications, 2020–2024.
- Keay N, Francis G, Lanfear M. Clinical application of monitoring indicators of female dancer health, including application of artificial intelligence in female hormone networks. Int J Sports Med Rehabil. 2022;5:24.
- Nix Biosensors. Real-time sweat electrolyte monitoring platform. Commercial product data, 2024–2025.
- Levels Health. CGM-based metabolic insights for athletes and health optimization. Platform data, 2023–2025.
- Function Health. Comprehensive biomarker panel (110+ markers including full hormonal panel). Service documentation, 2024–2025.
- Fountain Life. Executive health evaluation program including advanced hormonal profiling. Service documentation, 2024–2025.
- Catapult Sports. Athlete management systems and GPS-based tracking for professional sports teams. Company documentation, 2025.
- REDinSport / Health4Performance. Open-access educational resources for RED-S identification. Founded by Dr. Nicola Keay. https://www.health4performance.co.uk/
- Sundgot-Borgen J, Torstveit MK. Prevalence of eating disorders in elite athletes is higher than in the general population. Clin J Sport Med. 2004;14(1):25–32.
- Stellingwerff T, Mountjoy M, McCluskey WTP, Ackerman KE, Verhagen E, Heikura IA. Review of the scientific rationale, development and validation of the IOC REDs CAT2. Br J Sports Med. 2023;57(17):1109–1118.
- Hackney AC. A bad situation made worse: low carbohydrate intake amplifies low energy availability hormonal disturbances. BJSM Blog. August 2023.
- Joyner MJ, Coyle EF. Endurance exercise performance: the physiology of champions. J Physiol. 2008;586(1):35–44.
- Drinkwater BL, Nilson K, Chesnut CH III, Bremner WJ, Shainholtz S, Southworth MB. Bone mineral content of amenorrheic and eumenorrheic athletes. N Engl J Med. 1984;311(5):277–281.
- Ross E. Published work on sex-specific physiology in sports science at the English Institute of Sport. Multiple publications, 2018–2024.
- Sims ST, Heather AK. Myths and methodologies: reducing scientific design ambiguity in studies comparing sexes and/or menstrual cycle phases. Exp Physiol. 2018;103(10):1309–1317.
- Suzuki D, Suzuki Y. Identifying and analyzing low energy availability in athletes: the role of biomarkers and red blood cell turnover. Nutrients. 2024;16(14):2273.
#REDSinSport #LowEnergyAvailability #EnduranceAthletes #SportScience #WearableData #Biomarkers #Leptin #Cortisol #Testosterone #IGF1 #ThyroidHealth #Estradiol #DigitalTwin #LongevityPlanning #Healthspan #PrecisionMedicine #AIinSports #WearableTech #WHOOP #Oura #Garmin #FemaleAthlete #MaleAthlete #CorporateWellness #PredictiveHealth #IOC #SportNutrition #LongevityPlanAI #FunctionalMedicine #EnergyAvailability #TrainingLoad #HRV #HeartRateVariability #OvertrainingPrevention #BoneHealth #EndocrineHealth #PersonalizedMedicine