Peptide Science
·18 min read
The Wolverine Stack: Why Do BPC-157 and TB-500 Repair Injury So Well?
A science-first look at the two peptides everyone is stacking, the mechanisms that actually move the needle, and what personalized, data-driven medicine means for regenerative biology
By Tony Medrano, LongevityPlan.AI
The Wolverine Stack: Why Do BPC-157 and TB-500 Repair Injury So Well?
A science-first look at the two peptides everyone is stacking, the mechanisms that actually move the needle, and what personalized, data-driven medicine means for regenerative biology
By Tony Medrano | LongevityPlan.AI Research
The "Wolverine Stack" — BPC-157, a 15-amino-acid fragment isolated from human gastric juice by Predrag Sikirić in 1993, and TB-500, the LKKTETQ actin-binding fragment of thymosin β4. Together, these two peptides engage complementary repair pathways: angiogenesis and growth hormone receptor upregulation (BPC-157) and cell migration and stem cell mobilization (TB-500).
The 60-Second Brief
For the first time in modern sports medicine, two research peptides — BPC-157, a 15-amino-acid fragment isolated from human gastric juice by Dr. Predrag Sikirić in 1993, and TB-500, a synthetic version of the actin-binding region of thymosin β4 — have generated enough preclinical data to force a serious scientific conversation about what accelerated tissue repair means at the molecular level.12 The combined pair is sometimes nicknamed the "Wolverine Stack," after the Marvel character whose defining attribute is a regenerative healing factor.
This is a science-first article, not a protocol. We are going to do three things:
- Explain the actual biology — the pathways, the receptors, the gene expression cascades — that these two peptides engage, distilled from peer-reviewed literature as of 2026.
- Place that biology in the context of elite sports economics, military readiness, and the broader healthspan question that preventive medicine now has to answer: Can we bias tissue repair upward with targeted signaling molecules, and if so, for whom, when, and at what dose?
- Draw a clear, evidence-bounded line between what preclinical data supports, what human data does not yet support, and where the regulatory system (FDA, WADA) currently stands as of April 2026.
The punchline up front: the animal data for both peptides is broad, mechanistically coherent, and reproducible across many independent labs. The human data is extremely thin — three small pilot studies on BPC-157 (n=2 to n=16) and two Phase II trials on full-length thymosin β4 in chronic ulcers.34 Anyone telling you otherwise is not reading the same papers.
That gap — between rich preclinical signal and absent Phase III evidence — is exactly the kind of territory where AI-driven, data-rich personalized medicine has to go next. Digital twins, multi-omics phenotyping, and precision dose-finding are not luxuries here; they are the prerequisites for turning interesting biology into actual clinical tools.
Part I — Why This Matters Economically: The Cost of Soft Tissue Failure
Before we touch a single peptide, let's anchor the problem in numbers, because that's where LongevityPlan.AI's thesis always begins.
The NFL. The league estimates that more than 850 lower-body soft tissue strains occur each season, with hamstring strains being the single most common injury and the second-largest driver of missed play behind ACL tears.5 A 2024 longitudinal analysis in Orthopaedic Journal of Sports Medicine found a 33% reinjury rate for hamstring strains among NFL players — a statistic that has not meaningfully improved over a 12-year observation window.6 Injuries collectively cost NFL teams over $500 million in salaries paid to sidelined players in the 2019 season alone, according to Hospital for Special Surgery analysis reported by Forbes.7
The NBA. The 2023–24 season saw the highest rate of games missed to soft-tissue injury in the league's tracked history, putting hundreds of millions of dollars of payroll into non-productive cost. Stanford Athletics, the NBA's Phoenix Suns medical staff, and the US Olympic & Paralympic Committee's Performance Division have each identified time-to-return-to-competition as the single most valuable output metric they can optimize — more valuable than raw performance gains, because the expected-value math on availability dominates marginal strength or speed improvements.
The military. The US Army's Digital Twin initiative — which I've written about in a prior piece — identifies musculoskeletal injury as the single largest driver of non-combat medical attrition. The Department of Veterans Affairs estimates that over 70% of post-service disability claims involve a musculoskeletal component.
This is the addressable problem: tissue doesn't heal fast enough, doesn't heal cleanly, and scars out into weaker, stiffer, re-injury-prone replacements. The entire $213B global sports medicine and orthobiologics market is, in one sense, a very expensive workaround for the fact that adult human connective tissue has lousy innate healing kinetics compared to, say, a 10-year-old's or a salamander's.
So when two small peptides start showing up in peer-reviewed literature with effect sizes that consistently beat saline controls by 30–60% on histological, biomechanical, and functional endpoints in rat models of tendon, ligament, muscle, bone, and cornea — scientists pay attention, even if clinicians (correctly) insist on waiting for human trials.

Soft-tissue injury is the dominant cost driver in professional sports. Over 850 lower-body strains occur annually in the NFL, with hamstring strains carrying a 33% reinjury rate across a 12-year longitudinal cohort (Jenkins et al., Orthop J Sports Med, 2024). The $500M+ in 2019 sidelined-player salary cost (HSS/Forbes analysis) is the financial signal behind the NFL Scientific Advisory Board's $4M hamstring research initiative.
Part II — BPC-157: The Stomach Peptide That Isn't Really About the Stomach
Origin and Chemistry
BPC-157 is a 15-amino-acid fragment (sequence: Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val) derived from a larger protein called Body Protective Compound that Sikirić's group first isolated from human gastric juice in 1993.8 The peptide is unusually stable in the acidic environment of the stomach and in aqueous solution — a property that matters because most therapeutic peptides degrade rapidly in either context.9
It has had many names in the literature — PLD-116, PL-10, PL 14736, Bepectin — which is partly why a casual search turns up confusing results. It is the same molecule.
The Mechanism, Actually Explained
The best single resource on BPC-157's mechanism of action is the 2025 scoping review by McGuire, Martinez, Lenz, Skinner, and Cushman at the University of Utah Departments of Physical Medicine & Rehabilitation and Orthopaedics, published in Current Reviews in Musculoskeletal Medicine.10 I'm going to synthesize their framework here, because it's the most current and disciplined summary in the peer-reviewed literature.
Pathway 1: Angiogenesis via VEGFR2–Akt–eNOS. BPC-157 upregulates vascular endothelial growth factor receptor 2 (VEGFR2), activates the PI3K–Akt signaling axis, and turns on endothelial nitric oxide synthase (eNOS). The downstream effect is a measurable increase in nitric oxide production, vasodilation, and — critically for injury repair — new capillary formation at the wound site.1112 In a rat hind-limb ischemia model, Hsieh and colleagues at Chang Gung University (Taiwan) showed that BPC-157 both increased vessel density and accelerated blood flow recovery, with a dose-dependent phosphorylation of VEGFR2.13
This matters because tendons, ligaments, and the myotendinous junction are poorly vascularized by design. The reason Achilles ruptures take 6–12 months to fully remodel — and why corticosteroid injections can push that to 18 months — is that oxygen and growth factor delivery to the injury site is throttled by local capillary density. A molecule that upregulates VEGFR2 at the injury site is, in principle, solving exactly that rate-limiting step.
Pathway 2: ERK1/2 activation and EGR-1 transcription. BPC-157 dose-dependently phosphorylates the ERK1/2 MAP kinase cascade, which in turn drives expression of transcription factors c-Fos, c-Jun, and early growth response 1 (EGR-1).1415 When Huang et al. pharmacologically blocked the ERK pathway, BPC-157's pro-migratory and pro-angiogenic effects were abolished — a clean "necessary and sufficient" demonstration that establishes ERK1/2 as a non-negotiable node in the mechanism, not a correlate.16
Pathway 3: Src–caveolin-1–eNOS (the VEGF-independent branch). In 2020, Hsieh's group published a complementary paper in Scientific Reports showing that BPC-157 also activates eNOS through a Src kinase–caveolin-1 pathway that doesn't require VEGF.17 This is mechanistically important because it gives the peptide two redundant routes to the same endothelial protective output. Biology rarely builds redundancy without reason, and it helps explain why BPC-157's effects persist after the peptide itself has been cleared from plasma (half-life < 30 minutes).
Pathway 4: Growth hormone receptor upregulation in fibroblasts. Chang, Tsai, Hsu, and Pang's 2014 paper in Molecules — one of the primary references flagged for this article — demonstrated that BPC-157 increases growth hormone receptor (GHR) expression in cultured tendon fibroblasts, amplifying the anabolic response to circulating growth hormone.18 In plain language: BPC-157 makes your tendon cells "listen harder" to the growth hormone your body is already producing. This is a leveraged intervention — you're not adding growth hormone, you're increasing receptor density at the site that needs it.
Pathway 5: Anti-inflammatory cytokine modulation. Across rat models of colitis, periodontitis, and post-surgical wounds, BPC-157 reduces TNF-α, IL-6, and IFN-γ, and biases macrophage polarization from the pro-inflammatory M1 phenotype toward the reparative M2 phenotype.1920 M1-to-M2 macrophage switching is what separates acute productive inflammation from chronic fibrotic scarring — it is, arguably, the single most important transition in all of tissue repair biology.
Pathway 6: Neuromuscular junction stabilization. In rodent models, BPC-157 stabilizes acetylcholine receptor clustering at the neuromuscular junction and reverses paralysis induced by succinylcholine and lidocaine.2122 This is an underappreciated property — it means the peptide's effects aren't limited to connective tissue but extend to motor function after nerve or pharmacologic insult.
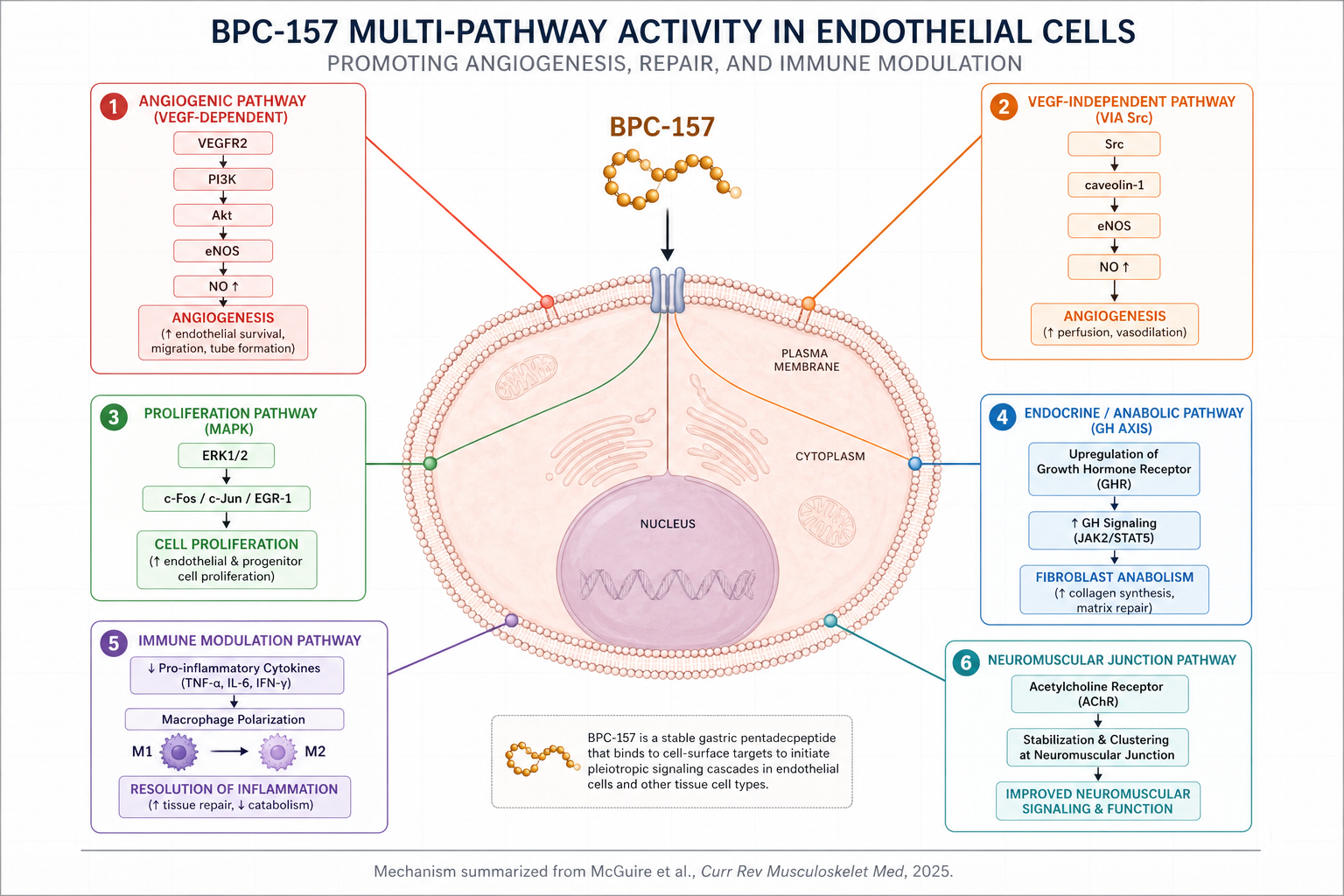
BPC-157 engages at least six overlapping molecular cascades, with VEGFR2-mediated angiogenesis and ERK1/2-driven transcription forming the mechanistic core. Inhibitor studies confirm that blocking the ERK pathway abolishes BPC-157's pro-angiogenic and pro-migratory effects — a necessary-and-sufficient demonstration that places ERK1/2 at the center of the repair program (Huang et al., Drug Des Devel Ther, 2015).
What The Animal Data Looks Like
Staresinic et al. (2003) transected rat Achilles tendons and showed that BPC-157-treated animals had significantly improved biomechanical load-to-failure and functional (Achilles Functional Index) recovery over 14 days compared to saline controls.23 Cerovecki et al. (2010) transected the medial collateral ligament in rats; the peptide-treated group showed faster collagen organization and improved tensile strength.24 Krivic et al. (2006) modeled Achilles detachment and demonstrated that BPC-157 promoted tendon-to-bone healing even in the presence of concurrent corticosteroid administration — a result that, if it held up in humans, would be clinically remarkable, because corticosteroid injections are a known inhibitor of tendon repair.25
Perhaps most striking: in a rat spinal cord injury model by Perovic et al., functional improvements and reduced spasticity persisted for 360 days after a single treatment course.26 The peptide's pharmacokinetic half-life is under 30 minutes. What's sustaining the effect is clearly not the peptide itself — it's the gene-expression cascade the peptide triggers within the first minutes of administration. BPC-157 appears to function less like a conventional drug and more like an epigenetic switch: a brief signal that produces a sustained biological response.
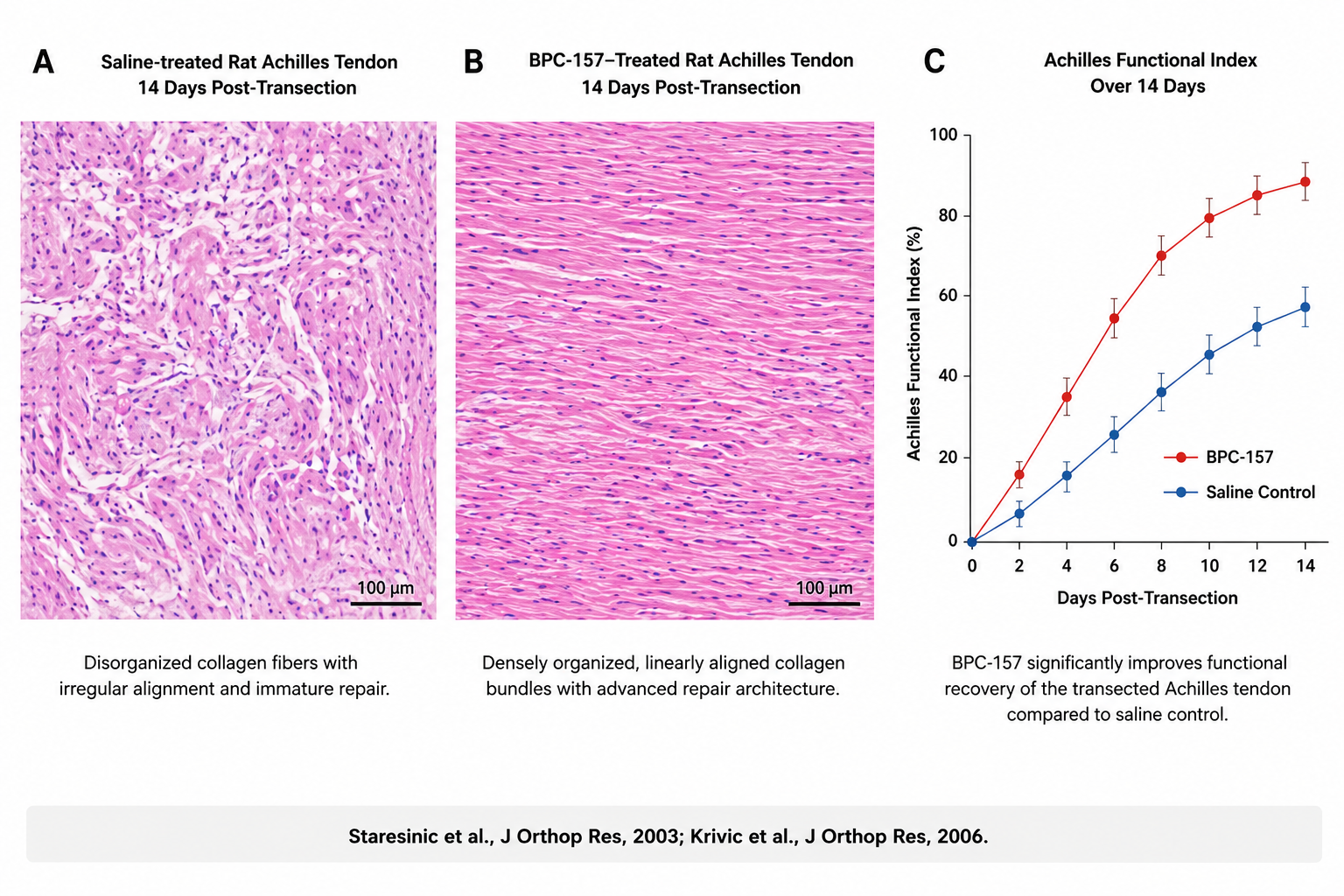
Histological comparison of saline-treated (A) versus BPC-157–treated (B) rat Achilles tendons 14 days after transection. The BPC-157 group shows densely organized, linearly aligned collagen bundles versus disorganized, irregular fiber alignment in controls. Functional recovery (C) is significantly faster across all 14 days post-injury (Staresinic et al., J Orthop Res, 2003; Krivic et al., J Orthop Res, 2006).
What The Human Data Actually Says
Three small pilot studies have been published, all from the Lee/Padgett group at Innate Healthcare:
- Lee & Padgett (2021): n=16 patients with knee pain receiving intra-articular BPC-157; significant pain reduction reported.27
- Lee et al. (2024): n=12 patients with interstitial cystitis; symptom improvement noted.28
- Lee & Burgess (2025): n=2 safety/feasibility study of intravenous BPC-157.29
A 2026 Yuan et al. review in International Journal of Molecular Sciences and a Józwiak et al. 2025 review both flag that a Phase I trial registered on ClinicalTrials.gov in 2015 has never reported results.3031 That is the complete state of human evidence. Three pilot studies, no randomized controlled trials, no Phase II or Phase III data.
Dr. Andrew Huberman covered BPC-157 in detail on his April 1, 2024 podcast, and his framing was scientifically careful: the animal data is interesting, the human data is preliminary, and any clinical use should be considered investigational.3233
The Cancer Question
The single most important safety question — and the one that gets the least serious treatment in the wellness press — is the theoretical cancer risk. Because BPC-157 upregulates VEGFR2, the same receptor that solid tumors hijack to vascularize themselves, there is a legitimate mechanistic concern about angiogenesis-mediated tumor promotion.
The published animal data is reassuring on this point. In a Kang et al. 2018 paper, BPC-157 actually reduced markers of cancer cachexia in a tumor-bearing mouse model, with no evidence of tumor growth acceleration.34 Sever et al. similarly found no pro-tumorigenic effects in a bile duct ligation model.35 But "no evidence in rats" is not "demonstrated safety in humans," and this is exactly the gap that proper clinical trials would close.
Part III — TB-500: The Actin-Binding Fragment That Mobilizes Stem Cells
Origin and Chemistry
TB-500 is the synthetic LKKTETQ fragment of thymosin β4 (Tβ4), a 43-amino-acid peptide originally isolated from calf thymus by Goldstein, Hannappel, Sosne, and Kleinman in the 1960s.36 The full-length Tβ4 molecule is one of the most abundant intracellular proteins in mammals. The LKKTETQ fragment, marketed as "TB-500," contains the actin-binding region and is administered as a research peptide for accelerated repair of muscle, tendon, and skin.
Important regulatory note: TB-500 (the fragment) and full-length Tβ4 are not the same molecule. Most clinical trial data is on full-length Tβ4. Both are now in FDA Category 2 of the 503A bulks list.37
Mechanism of Action
Tβ4's primary biological role is to sequester monomeric G-actin, the building block of the cytoskeleton. By binding G-actin, it regulates actin polymerization — the dynamic process that drives every form of cell movement, including the migration of fibroblasts, keratinocytes, endothelial cells, and stem cells into a wound bed.38
The downstream consequences of this actin regulation are remarkably broad:
- Endothelial cell migration and tube formation. Tβ4 promotes the migration of vascular endothelial cells and the formation of capillary-like structures in vitro.39
- Cardiomyocyte progenitor recruitment. In a landmark 2004 Nature paper, Bock-Marquette and colleagues showed that intracardiac Tβ4 administration promoted myocyte survival and recruited epicardial progenitor cells after experimental myocardial infarction.40
- Anti-inflammatory and anti-apoptotic signaling. Tβ4 suppresses NF-κB activation, reducing the downstream inflammatory cytokine cascade. In corneal injury models, this translates to faster re-epithelialization with less scarring.41
- Myofibroblast reduction — less scar, more function. This is the mechanism that makes Tβ4 particularly interesting for tendon and muscle repair. Tβ4 decreases the number of myofibroblasts that form at a wound site, which translates directly to reduced fibrotic scar tissue. In an MCL model, Tβ4 delivered in fibrin sealant produced "uniform, evenly spaced collagen fiber bundles at 4 weeks" — a histological finding that maps directly onto better biomechanical recovery.42
The Human Clinical Data — Actually Better Than BPC-157
This is where TB-500's parent molecule, full-length Tβ4, has a genuine edge. RegeneRx Biopharmaceuticals (the company developed by collaborators of Allan Goldstein, who first isolated thymosin from calf thymus in the 1960s) ran two Phase II clinical trials of Tβ4 in patients with chronic venous stasis ulcers and pressure ulcers. As reported by Treadwell, Kleinman, Crockford, Hardy, Guarnera, and Goldstein in the Annals of the New York Academy of Sciences in 2012, Tβ4 "accelerated healing by almost a month in those patients that did heal."43 In rat full-thickness wound models, topical or intraperitoneal Tβ4 increased re-epithelialization by 42% at 4 days and 61% at 7 days versus saline controls (Malinda et al., 1999).44
The ocular program, marketed as RGN-259, advanced to Phase 3 trials for neurotrophic keratopathy and Phase 2 for dry eye disease, with published dermal wound healing data in nine patients with chronic non-healing neurotrophic corneal epithelial defects.
Note carefully: these human trials were done with full-length Tβ4, not TB-500. The extrapolation from full Tβ4 human data to TB-500 (the LKKTETQ fragment) is a mechanistic inference, not a demonstrated clinical equivalence.
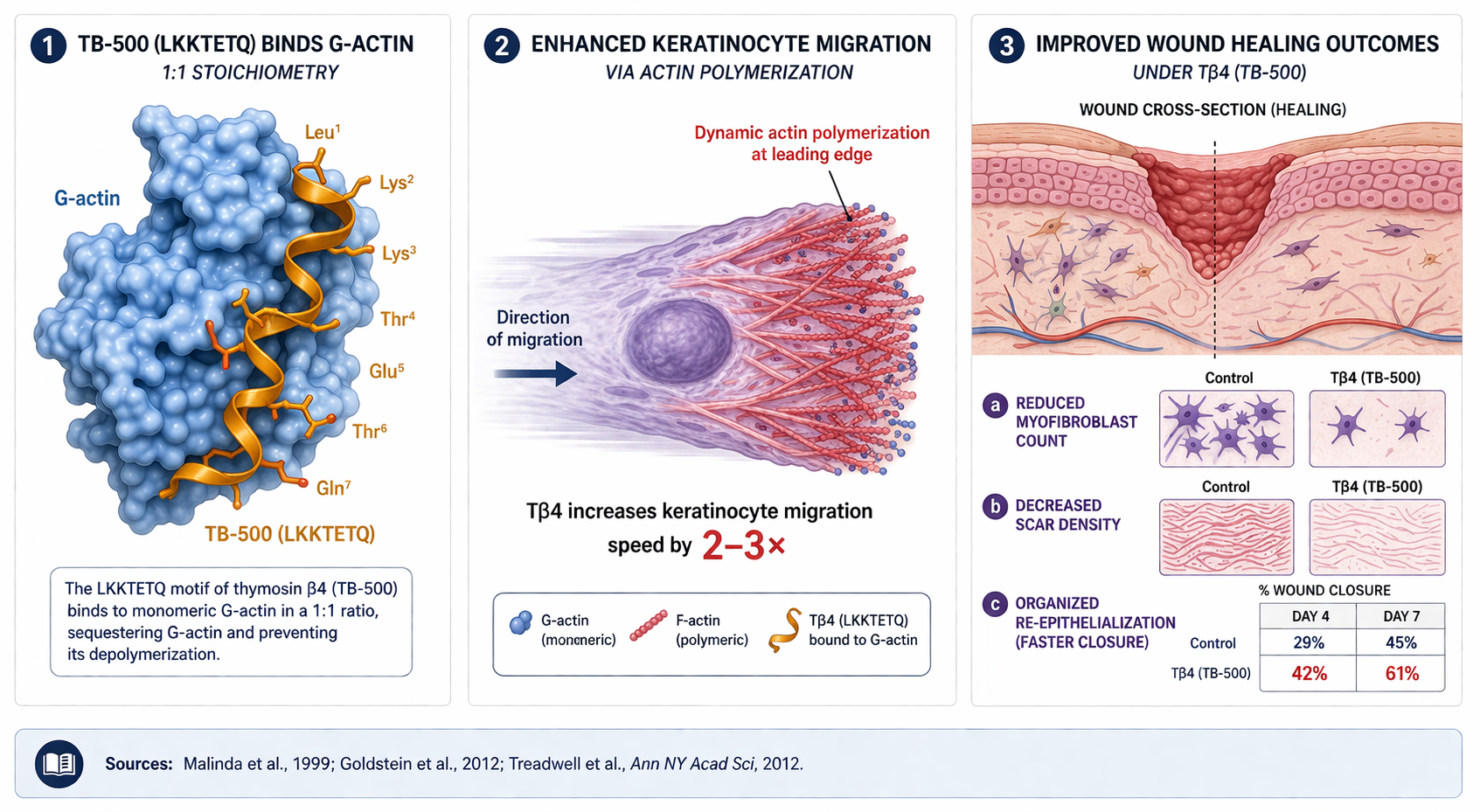
TB-500's core mechanism is deceptively simple: bind G-actin, regulate the monomer pool, and accelerate cell migration. Downstream, this produces 42% greater re-epithelialization at day 4 and 61% at day 7 in rat full-thickness wound models (Malinda et al., 1999), with reduced myofibroblast infiltration — meaning less fibrotic scar and more functional repair. Two Phase II trials of full-length thymosin β4 in chronic ulcers showed accelerated healing by roughly one month in patients who responded.
Part IV — Why "Stacking" Them Makes Biological Sense (and Why the Evidence Base Still Lags the Logic)
Here's the steel-man argument for why athletes and sports medicine clinicians started pairing the two peptides: the mechanisms are complementary, not redundant.
| Axis | BPC-157 | TB-500 / Tβ4 |
|---|---|---|
| Primary biology | Angiogenesis, growth hormone receptor upregulation, NO signaling | Actin sequestration, cell migration, stem cell mobilization |
| Strongest tissue | Tendon, ligament, bone, gut mucosa | Skin, cornea, cardiac muscle, skeletal muscle |
| Delivery mode in literature | Local or systemic; stable in gastric fluid | Systemic (circulates to injury sites) |
| Primary mechanism duration | Gene-expression switch; effects persist weeks–months after clearance | Continuous during administration; shorter tail |
| Human evidence | 3 pilot studies, n ≤ 16 per study | 2 Phase II trials (Tβ4 full length) + Phase 2/3 ocular |
| Inflammation pathway | TNF-α, IL-6, IFN-γ reduction; M1→M2 macrophage shift | NF-κB suppression; myofibroblast reduction |
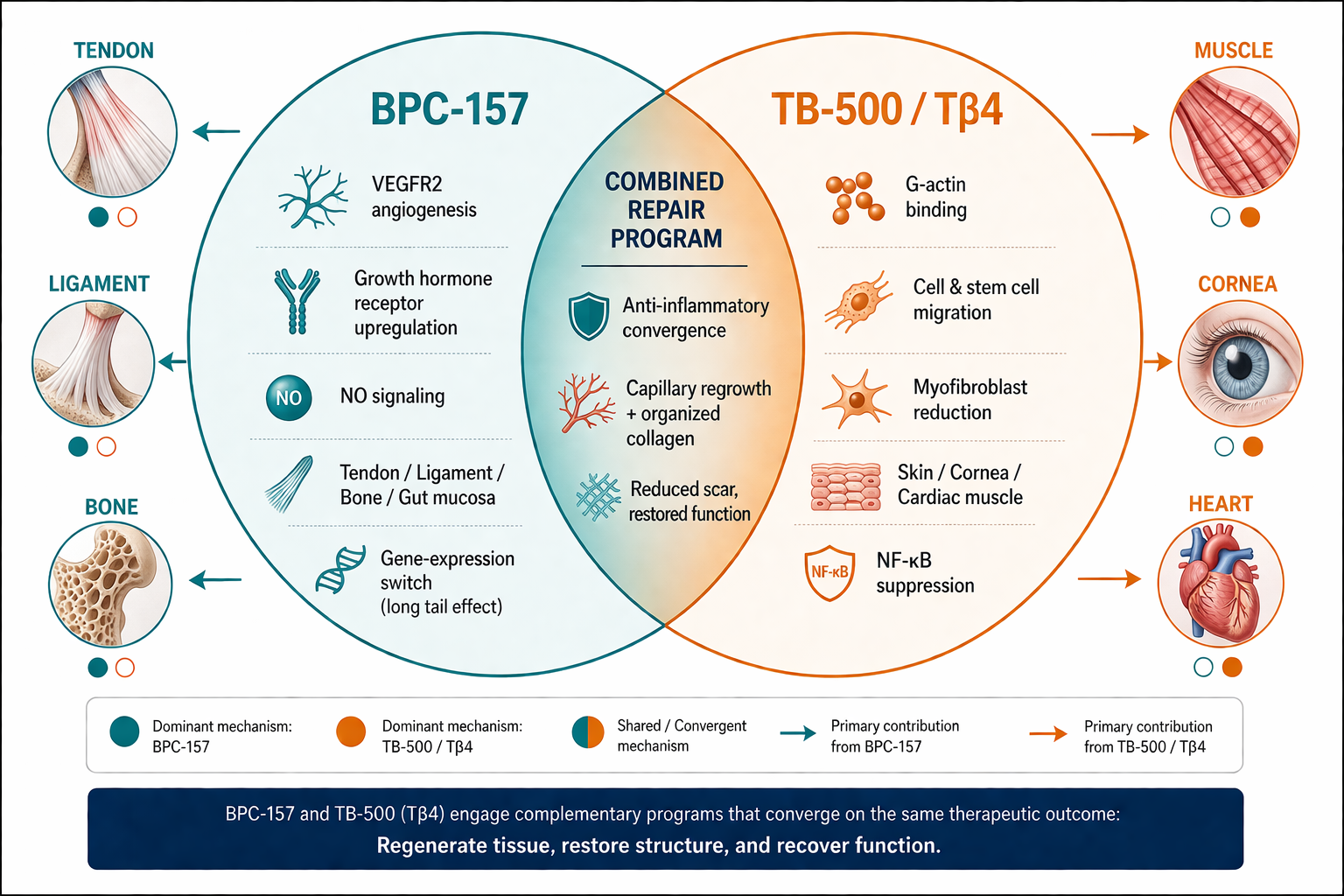
BPC-157 and TB-500 (Tβ4) engage complementary, non-overlapping repair programs that converge on the same therapeutic outcome: regenerate tissue, restore structure, and recover function. BPC-157 dominates the angiogenic, growth-hormone, and NO-signaling axes (favoring tendon, ligament, bone, gut). TB-500 dominates G-actin–mediated cell migration and myofibroblast reduction (favoring skin, cornea, cardiac and skeletal muscle). The shared anti-inflammatory zone is where the "stack" hypothesis lives.
A tendon-to-bone reattachment, for instance, requires both local neovascularization (BPC-157's wheelhouse) and organized collagen fiber deposition with minimal scarring (Tβ4's wheelhouse). A myocardial infarction requires both capillary regrowth and cardiomyocyte preservation — again, complementary. So the biological logic of combining them is sound.
But — and this is the LongevityPlan.AI standard — biological logic is not evidence. There is no published human trial of the BPC-157 + TB-500 combination. The Lee & Padgett 2021 study included four patients who received BPC-157 plus thymosin β4 for knee pain; three reported significant improvement, one did not.45 That is the entirety of the controlled combination data in humans.
When a practitioner tells you "the stack works better than either alone," they are extrapolating from mechanism, not citing a trial. That distinction is worth keeping.
Part V — The Regulatory and Scientific State of Play, April 2026
The regulatory landscape matters, because it shapes both who can access these compounds and what research gets done.
WADA has prohibited BPC-157 under the S0 (Unapproved Substances) category since January 1, 2022, in and out of competition.46 TB-500 and related fragments are prohibited under S2 (Peptide Hormones, Growth Factors) on the 2025 WADA list. Any elite athlete using these compounds is subject to sanction.
FDA placed BPC-157, TB-500 (LKKTETQ fragment), and approximately 20 other peptides into Category 2 of the 503A compounding bulks list on September 29, 2023, citing "significant safety risks" — specifically, immunogenicity concerns, manufacturing impurities, and the absence of human safety data.4748 Category 2 placement effectively prohibits US compounding pharmacies from preparing these substances for human use.
Recent development. In April 2026, following nominator withdrawals, FDA removed BPC-157 and 11 other peptides from Category 2 status with the announcement that the Pharmacy Compounding Advisory Committee (PCAC) would review them at the July 23, 2026 meeting.49 This is not an approval — it is a procedural reset that opens the door to a formal scientific review. The outcome of that meeting will shape the legal compounding landscape for both peptides.
DOJ enforcement. The Department of Justice has taken enforcement action — Tailor Made Compounding LLC was required to forfeit $1.79 million for distributing unapproved peptides including BPC-157.50 Clinics and telehealth platforms should regard this compound as genuinely unapproved, not as a gray-area wellness supplement.
Part VI — Why Science and Data Matter More Here Than Almost Anywhere Else
This is where LongevityPlan.AI's core thesis — that preventive medicine has to be personalized, data-rich, and AI-augmented — actually earns its keep. Consider the clinical decision space for someone with a tendon injury who is asking whether a peptide protocol makes sense for them:
Dimension 1: Baseline angiogenic capacity. Individuals vary enormously in baseline VEGF expression, endothelial function, and NO bioavailability. An aging executive with subclinical endothelial dysfunction has a different starting point than a 24-year-old Olympic gymnast. Measuring circulating VEGF, asymmetric dimethylarginine (ADMA, an eNOS inhibitor), and flow-mediated dilation gives a meaningful baseline.
Dimension 2: Injury phenotype. A partial hamstring strain at the myotendinous junction is mechanistically different from a full Achilles rupture. A stress fracture with compromised vascular supply is different from an acute traumatic tibial fracture. Quantitative MRI, T2 mapping, and ultrasound elastography can phenotype an injury in ways that didn't exist even a decade ago.
Dimension 3: Cancer risk profile. Because BPC-157 upregulates VEGFR2, individuals with known cancer history, heritable cancer syndromes (BRCA1/2, Lynch syndrome), or elevated circulating tumor DNA should be approached with extreme caution. This is exactly the kind of patient-level contraindication that a properly structured digital twin would flag before the clinical conversation even begins.
Dimension 4: Drug interactions. BPC-157's stimulation of nitric oxide signaling has theoretical interactions with nitrate medications, phosphodiesterase-5 inhibitors, and antihypertensive regimens. The pharmacokinetic interplay has not been characterized in humans.
Dimension 5: Dose-finding. Dr. Huberman has publicly discussed the typical clinically-prescribed range of 300–500 micrograms subcutaneously, two to three times per week, for an 8-week course.51 This is an inherited heuristic, not a dose that has been validated by randomized trials. The toxicity data is reassuring — Xu et al. (2020) could not identify a minimum toxic dose or lethal dose in animal models, with no teratogenic, genotoxic, anaphylactic, or local toxic effects — but "no observed toxicity" is not "optimal efficacy," and the difference is exactly what dose-ranging trials are supposed to characterize.52
This is the scientific case for digital twin modeling, multi-omics phenotyping, and AI-driven protocol personalization of the kind that the Buck Institute, the Stanford Center on Longevity, the Salk Institute, and MIT's Langer Lab have been pushing toward for a decade. Dr. Robert Langer — the most-cited engineer in history with over 472,000 citations and the founder of the modern field of tissue engineering — has spent his career building precisely the controlled-release and targeted-delivery systems that would let a molecule like BPC-157 actually be studied with the precision it deserves.53 His work is a reminder that "peptide therapy" without a delivery system is like a drug without a pill — the pharmacology is only as good as the formulation.
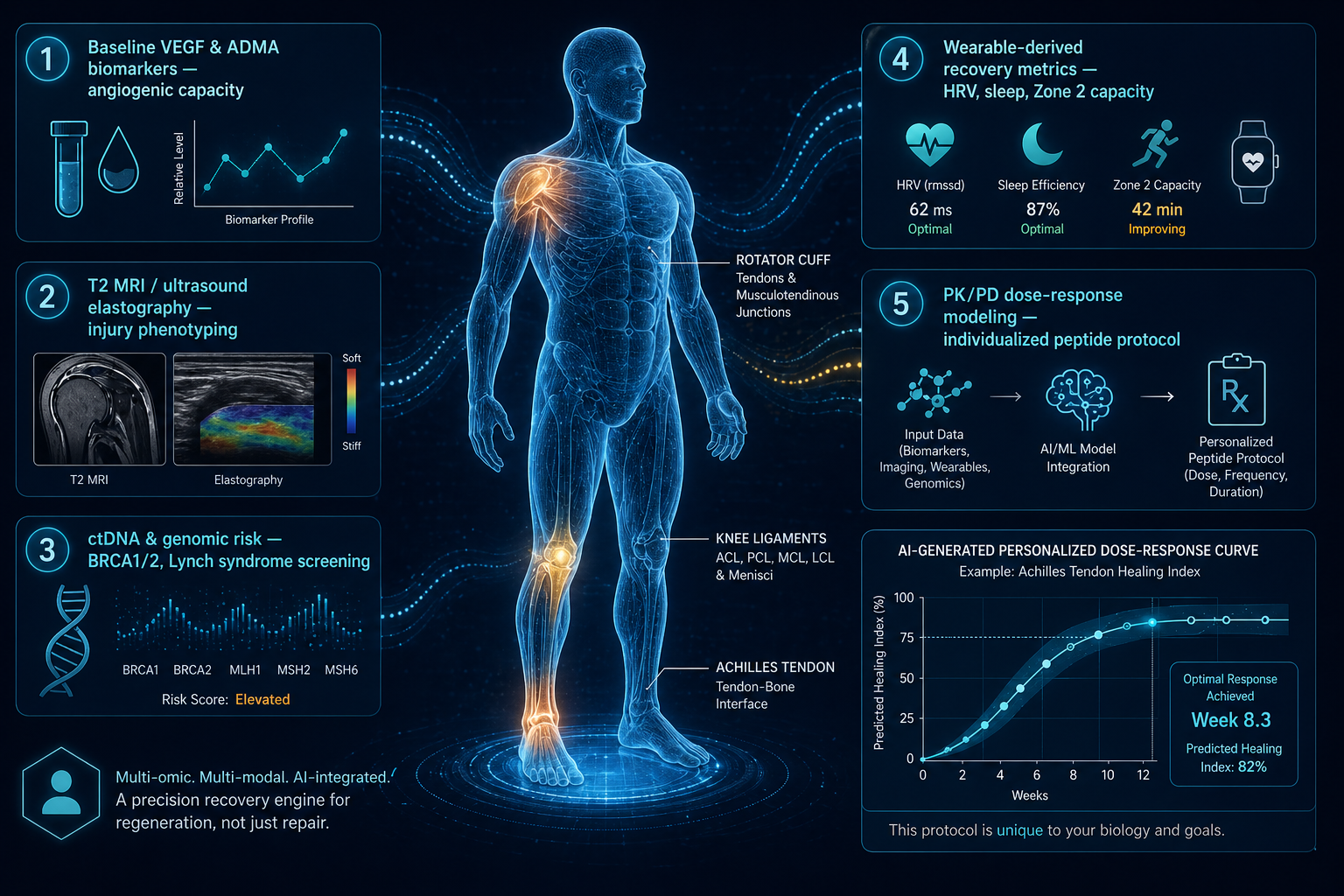
Peptide therapy without personalization is just peptide roulette. A properly structured digital twin integrates baseline endothelial function, angiogenic biomarkers (VEGF, ADMA), genomic cancer risk, injury phenotype, and wearable-derived recovery data to generate patient-specific dose-response predictions. This is the operational framework that Insilico Medicine, Recursion Pharmaceuticals, PathAI, and the Langer Lab at MIT are progressively making clinically feasible — and the only credible path for translating mechanistically promising peptides like BPC-157 and TB-500 into rigorous, responsible clinical use.
Part VII — What Would a Real Clinical Research Program Look Like?
If I were advising a pharma sponsor or an NIH extramural program on what a credible clinical development program for BPC-157 or the BPC-157 + TB-500 combination should look like, it would have five components:
-
A properly-powered Phase I/II dose-escalation in a single indication. Achilles tendinopathy and chronic non-healing wounds are the two best candidates based on mechanistic fit and existing preclinical data. n = 150 per arm, three dose levels, placebo control, with MRI-based primary endpoints.
-
A cancer surveillance arm. Given the VEGFR2 concern, any trial needs CT-DNA monitoring, whole-body MRI screening at baseline and follow-up, and rigorous exclusion of patients with any oncologic history.
-
Independent replication of Sikirić's core findings. The IJMS review by Yuan and colleagues at Johns Hopkins explicitly flagged that a considerable portion of the current evidence arises from a single research group. That is an honest and appropriate concern. Independent replication by Stanford's Institute for Stem Cell Biology and Regenerative Medicine, the Buck Institute, the Harvard Stem Cell Institute, or the Salk Institute would meaningfully change the confidence interval on the mechanistic claims.
-
Pharmacokinetic/pharmacodynamic (PK/PD) modeling that explains the half-life paradox. A molecule with a 30-minute plasma half-life producing 360-day functional improvements (Perovic et al., 2019) is not a conventional pharmacological profile. That gap is where the most important mechanistic science lives — it probably involves epigenetic reprogramming or self-sustaining paracrine loops, both of which are testable.54
-
A digital twin framework for patient stratification. Not every patient with a rotator cuff tear will respond the same way to the same peptide protocol. The baseline endothelial function, inflammatory tone, age, sex, hormonal status, and specific injury phenotype all matter. A properly structured digital twin that integrates wearable data, genomic risk scores, and serial biomarker panels could let a single trial generate effectively personalized dose-response curves — the kind of work that Insilico Medicine, Recursion Pharmaceuticals, and PathAI are beginning to make operationally possible.
This is not a speculative wish list. Every component of it is being executed in neighboring fields. The peptide regeneration field has been unusually slow to adopt modern clinical trial methodology, and that is exactly why the evidence gap has stayed open for 30 years.
Part VIII — The Bottom Line
What we know with high confidence:
- BPC-157 and TB-500/Tβ4 both activate coherent, well-characterized molecular pathways that plausibly accelerate tissue repair.
- Animal data across tendon, ligament, muscle, bone, cornea, cardiac, and gastrointestinal injury models is broad, replicated (for Tβ4, across multiple independent labs; for BPC-157, largely within Sikirić's group and allied labs), and mechanistically consistent.
- Toxicity signals in animal models are, to date, minimal. Xu et al. could not establish an LD50 or minimum toxic dose for BPC-157. Full-length Tβ4 has gone through Phase II human trials without major safety issues.
What we do not know:
- Whether the observed effect sizes in rodents translate to clinically meaningful outcomes in humans.
- The optimal dose, route, duration, and cycling schedule for any human indication.
- The long-term cancer risk profile, particularly for BPC-157 given its VEGFR2 agonist activity.
- Whether the BPC-157 + TB-500 combination produces additive, synergistic, or null benefit over either alone.
What the regulatory system says:
- Both peptides are WADA-prohibited for competitive athletes.
- Both are non-approved, with FDA compounding status under PCAC review as of July 2026.
- Current distribution channels are largely unregulated "research chemical" sources of uncertain purity and dosage accuracy.
What this means for the LongevityPlan.AI thesis:
The Wolverine Stack is not a wellness hack, and it is not a scam. It is a genuinely interesting pair of signaling molecules operating at the frontier of regenerative biology, badly in need of the kind of rigorous, personalized, data-intensive clinical investigation that modern AI-driven medicine is uniquely equipped to deliver. The gap between mechanistic promise and clinical evidence is not a problem you solve by shouting louder on Instagram — it is a problem you solve by running properly designed trials in properly phenotyped patients with properly measured endpoints.
That is the work. And that is the only way tissue regeneration ever gets from rodent models to routine clinical practice.
About the Author

Tony Medrano is CEO and Founder of LongevityPlan.AI, a company that integrates Peptide Therapy and luxurious, fun, and athletic monthly virtual wellness retreats for busy individuals, teams, and corporations on the go with the latest in Performance Science Research to Optimize for Physical, Mental, and Aesthetic Longevity.
LongevityPlan serves all 50 US States with American-made, US Doctor-prescribed, FDA-approved peptides that are shipped discreetly from US 503A Pharmacies directly to the doorsteps of its elite clients.
LongevityPlan's platform integrates performance and health data from its sophisticated clients and leverages its proprietary Digital Twin for Predictive Peptide Performance™ technology, wearable data, biomarker data, EMR data, and coach-patient conversations to deliver personalized performance optimization and longevity recommendations to athletes, coaches, organizations, businesses, government, and the military.
In addition to being a 3x technology / AI company CEO with 2 successful exits, Tony has also finished 3 Full Ironman Triathlons (140.6 mi) since 2019. He has degrees from Harvard University, Columbia University, and a JD/MBA from Stanford University. Tony also served as a US Navy Officer commanding an emergency response team on a USN Destroyer during counter-narcotics operations in Latin America.
Follow the newsletter for deep-dive analyses on AI-driven preventive medicine, digital twin technology, and the science of healthspan.
#Peptides #BPC157 #TB500 #RegenerativeMedicine #SportsMedicine #Longevity #Healthspan #DigitalTwin #PreventiveMedicine #SportsInjury #TendonRepair #Angiogenesis #HubermanLab #WADA #FDA #LongevityPlanAI
References
Footnotes
-
Sikirić P, Petek M, Ručman R, et al. A new gastric juice peptide, BPC. An overview of the stomach-stress-organoprotection hypothesis and beneficial effects of BPC. J Physiol (Paris). 1993;87:313–327. PMID: 8298609. ↩
-
Goldstein AL, Hannappel E, Sosne G, Kleinman HK. Thymosin β4: a multi-functional regenerative peptide. Basic properties and clinical applications. Expert Opinion on Biological Therapy. 2012. PMID: 22074294. ↩
-
McGuire FP, Martinez R, Lenz A, Skinner L, Cushman DM. Regeneration or Risk? A Narrative Review of BPC-157 for Musculoskeletal Healing. Curr Rev Musculoskelet Med. 2025;18(12):611–619. doi:10.1007/s12178-025-09990-7. PMCID: PMC12446177. ↩
-
Yuan C, Demers A, Silva-Ortiz V, Hasoon JJ, Lee W, Dave K, Amirdelfan K, Burke HW, Christo PJ, Robinson CL. From Regeneration to Analgesia: The Role of BPC-157 in Tissue Repair and Pain Management. Int J Mol Sci. 2026;27(6):2876. doi:10.3390/ijms27062876. ↩
-
NFL Scientific Advisory Board, NFL Player Health and Safety. Lower Body Soft Tissue Strain Research Program. https://www.nfl.com/playerhealthandsafety ↩
-
Jenkins R, et al. Risk and Prognosis of Hamstring Injuries in the National Football League: A 12-Year Review. Orthop J Sports Med. 2024 Dec 6;12(12):23259671241298622. PMCID: PMC11624564. ↩
-
Hospital for Special Surgery, as reported in Forbes, February 2020: "Injuries Cost NFL Teams Over $500 Million in 2019." ↩
-
Sikirić P, Petek M, Ručman R, et al. J Physiol (Paris). 1993;87:313–327. PMID: 8298609. ↩
-
Pflaum Z, Ručman R. Solid Phase Peptide Synthesis of the Fragment BPC 157 of Human Gastric Juice Protein BPC and Its Analogues. Acta Chim Slov. 2005;52:34–39. ↩
-
McGuire FP, et al. Curr Rev Musculoskelet Med. 2025;18(12):611–619. PMCID: PMC12446177. ↩
-
McGuire FP, et al. Curr Rev Musculoskelet Med. 2025;18(12):611–619. PMCID: PMC12446177. ↩
-
Hsieh MJ, Liu HT, Wang CN, et al. Therapeutic potential of pro-angiogenic BPC157 is associated with VEGFR2 activation and up-regulation. J Mol Med (Berl). 2017;95:323–333. PMID: 27847966. ↩
-
Hsieh MJ, et al. J Mol Med (Berl). 2017;95:323–333. PMID: 27847966. ↩
-
Huang T, Zhang K, Sun L, et al. Body protective compound-157 enhances alkali-burn wound healing in vivo and promotes proliferation, migration, and angiogenesis in vitro. Drug Des Devel Ther. 2015;9:2485–2499. PMCID: PMC4425239. ↩
-
Tkalčević VI, Čužić S, Brajša K, et al. Enhancement by PL 14736 of granulation and collagen organization in healing wounds and the potential role of egr-1 expression. Eur J Pharmacol. 2007;570:212–221. PMID: 17628536. ↩
-
Huang T, et al. Drug Des Devel Ther. 2015;9:2485–2499. PMCID: PMC4425239. ↩
-
Hsieh MJ, Lee CH, Chueh HY, et al. Modulatory effects of BPC 157 on vasomotor tone and the activation of Src-Caveolin-1-endothelial nitric oxide synthase pathway. Sci Rep. 2020;10:17078. PMCID: PMC7555539. ↩
-
Chang CH, Tsai WC, Hsu YH, Su Pang JH. Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts. Molecules. 2014;19:19066–19077. PMCID: PMC6271067. ↩
-
McGuire FP, et al. Curr Rev Musculoskelet Med. 2025;18(12):611–619. PMCID: PMC12446177. ↩
-
Kang EA, Han YM, An JM, et al. BPC157 as potential agent rescuing from cancer cachexia. Curr Pharm Des. 2018;24:1947–1956. PMID: 29898649. ↩
-
McGuire FP, et al. Curr Rev Musculoskelet Med. 2025;18(12):611–619. PMCID: PMC12446177. ↩
-
Sikiric P, Boban Blagaic A, Strbe S, et al. The stable gastric pentadecapeptide BPC 157 pleiotropic beneficial activity and its possible relations with neurotransmitter activity. Pharmaceuticals (Basel). 2024;17:461. PMCID: PMC11053547. ↩
-
Staresinic M, Sebecic B, Patrlj L, et al. Gastric pentadecapeptide BPC 157 accelerates healing of transected rat Achilles tendon and in vitro stimulates tendocytes growth. J Orthop Res. 2003;21:976–983. PMID: 14554208. ↩
-
Cerovecki T, Bojanic I, Brcic L, et al. Pentadecapeptide BPC 157 (PL 14736) improves ligament healing in the rat. J Orthop Res. 2010;28:1155–1161. PMID: 20225319. ↩
-
Krivic A, Anic T, Seiwerth S, Huljev D, Sikiric P. Achilles detachment in rat and stable gastric pentadecapeptide BPC 157: promoted tendon-to-bone healing and opposed corticosteroid aggravation. J Orthop Res. 2006;24:982–989. PMID: 16583442. ↩
-
Perovic D, Kolenc D, Bilic V, et al. Stable gastric pentadecapeptide BPC 157 can improve the healing course of spinal cord injury and lead to functional recovery in rats. J Orthop Surg Res. 2019;14:199. PMCID: PMC6604284. ↩
-
Lee E, Padgett B. Intra-Articular Injection of BPC 157 for Multiple Types of Knee Pain. Altern Ther Health Med. 2021;27. PMID: 34324435. ↩
-
Lee E, Walker C, Ayadi B. Effect of BPC-157 on symptoms in patients with interstitial cystitis: A pilot study. Altern Ther Health Med. 2024;30:12–17. PMID: 39325560. ↩
-
Lee E, Burgess K. Safety of Intravenous Infusion of BPC157 in Humans: A Pilot Study. Altern Ther Health Med. 2025. PMID: 40131143. ↩
-
Yuan C, et al. Int J Mol Sci. 2026;27(6):2876. doi:10.3390/ijms27062876. ↩
-
ClinicalTrials.gov listing, 2015 BPC-157 Phase I trial; results not reported per Yuan et al. 2026 and Józwiak et al. 2025 reviews. ↩
-
Huberman A. Benefits & Risks of Peptide Therapeutics for Physical & Mental Health. Huberman Lab Podcast, April 1, 2024. ↩
-
Huberman A. Huberman Lab Podcast, April 1, 2024. ↩
-
Kang EA, et al. Curr Pharm Des. 2018;24:1947–1956. ↩
-
Sever AZ, Sever M, Vidovic T, et al. Stable gastric pentadecapeptide BPC 157 in the therapy of the rats with bile duct ligation. Eur J Pharmacol. 2019;847:130–142. PMID: 30690000. ↩
-
Goldstein AL, Hannappel E, Sosne G, Kleinman HK. Thymosin β4: a multi-functional regenerative peptide. Expert Opinion on Biological Therapy. 2012;12(1):37–51. ↩
-
FDA Updated 503A Bulks List Categories, September 29, 2023. Thymosin Beta-4, Fragment (LKKTETQ) and BPC-157 added to Category 2. ↩
-
Goldstein AL, et al. Expert Opin Biol Ther. 2012;12(1):37–51. ↩
-
Malinda KM, Sidhu GS, Mani H, et al. Thymosin β4 accelerates wound healing. J Invest Dermatol. 1999;113:364–368. PMID: 10469335. ↩
-
Bock-Marquette I, Saxena A, White MD, Dimaio JM, Srivastava D. Thymosin β4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair. Nature. 2004;432:466–472. ↩
-
Sosne G, Qiu P, Christopherson PL, Wheater MK. Thymosin beta 4 suppression of corneal NFκB: a potential anti-inflammatory pathway. Exp Eye Res. 2007;84:663–669. ↩
-
Malinda KM, et al. J Invest Dermatol. 1999;113:364–368. PMID: 10469335. ↩
-
Treadwell T, Kleinman HK, Crockford D, Hardy MA, Guarnera GT, Goldstein AL. The regenerative peptide thymosin β4 accelerates the rate of dermal healing in preclinical animal models and in patients. Ann N Y Acad Sci. 2012;1270:1306–1310. PMID: 23050815. ↩
-
Malinda KM, Goldstein AL, Kleinman HK. Thymosin β4 stimulates directional migration of human umbilical vein endothelial cells. FASEB J. 1997;11:474–481. ↩
-
Lee E, Padgett B. Altern Ther Health Med. 2021;27. PMID: 34324435. ↩
-
USADA / WADA 2022 Prohibited List; BPC-157 prohibited under S0 Unapproved Substances, effective January 1, 2022. ↩
-
FDA Updated 503A Bulks List Categories, September 29, 2023. ↩
-
Alliance for Pharmacy Compounding correspondence, October 27, 2023; FDA Office of Compounding Quality and Compliance correspondence. ↩
-
FDA announcement, April 2026, removal of BPC-157 and 11 other peptides from Category 2; PCAC review scheduled July 23, 2026. ↩
-
US Department of Justice action against Tailor Made Compounding LLC, reported in DJ Holt Law peptide regulatory analysis, 2025–2026. ↩
-
Huberman A. Huberman Lab Podcast, April 1, 2024. ↩
-
Xu C, Sun L, Ren F, et al. Preclinical safety evaluation of body protective compound-157, a potential drug for treating various wounds of tendon and tissue. Regul Toxicol Pharmacol. 2020;114:104665. ↩
-
Langer R. Chemical and biological approaches to regenerative medicine and tissue engineering. Molecular Frontiers Journal. 2019;3(2). Langer Lab, MIT. https://langerlab.mit.edu/ ↩
-
Perovic D, et al. J Orthop Surg Res. 2019;14:199. PMCID: PMC6604284. ↩