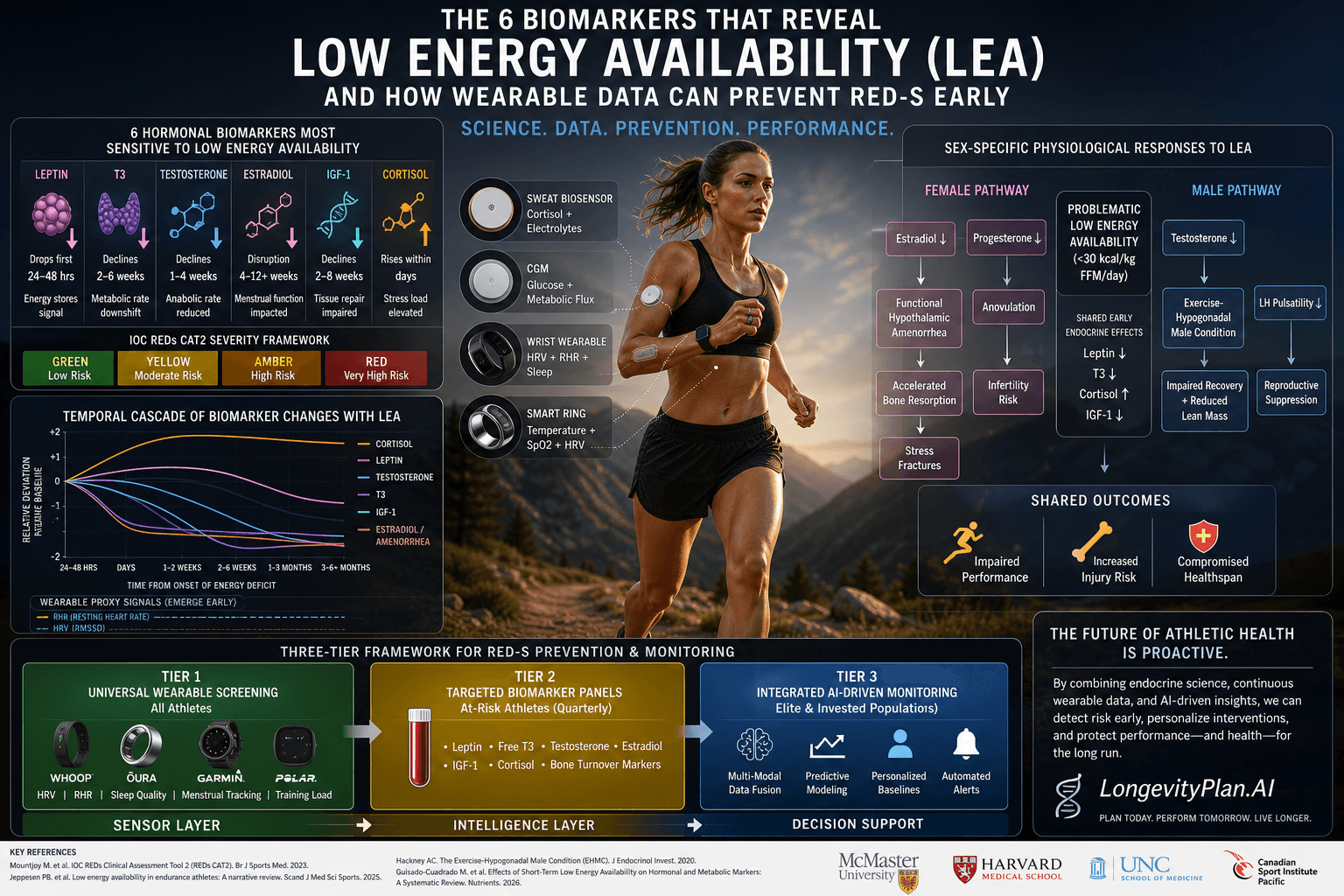
Peptide Science
·14 min read
Five Peptides Every Endurance Athlete Should Understand
From Mitochondrial Signaling to Connective-Tissue Remodeling—and How AI Helps
By Tony Medrano, LongevityPlan.AI

Five Peptides Every Endurance Athlete Should Understand—From Mitochondrial Signaling to Connective-Tissue Remodeling—and How AI Helps
By Tony Medrano | CEO & Co-Founder | LongevityPlan.AI | March 2026
Introduction: Why Peptides Are the New Frontier for Endurance Athletes
In the spring of 2015, a small laboratory at the USC Leonard Davis School of Gerontology announced something that made exercise physiologists sit up straight: a sixteen-amino-acid molecule encoded not in our familiar nuclear DNA but in our mitochondrial genome could, when injected into obese mice, replicate many metabolic benefits of exercise—suppressing weight gain, reversing insulin resistance, and boosting endurance. The molecule was called MOTS-c, and its discoverers—Changhan David Lee and Pinchas Cohen—had effectively opened a door that the endurance community has been walking through ever since.
Six years later, Reynolds et al. published a landmark study in Nature Communications showing that MOTS-c treatment could double treadmill running capacity in old mice, restoring physical performance to levels seen in animals a fraction of their age. Meanwhile, orthopedic surgeons were cataloguing a parallel revolution: synthetic peptides like BPC-157, originally derived from a protective protein in human gastric juice, were showing preclinical promise in accelerating tendon, ligament, and muscle repair—the very tissues endurance athletes stress on an almost daily basis.
The peptide therapeutics market is no longer a niche curiosity. As of 2024, roughly 60 FDA-approved peptide medications exist, 140 are in active clinical trials, and more than 500 are in preclinical development. Google search volumes for compounds like BPC-157 and MOTS-c hit all-time highs in mid-2024, with more than 50 million tagged video views across YouTube and TikTok. The interest is not merely recreational: endurance coaches and practitioners at organizations from the US Olympic Committee to Stanford Athletics are paying close attention, because the intersection of peptide science, AI-driven drug discovery, and wearable-data analytics is producing insights that could reshape how we train, recover, and extend competitive careers.
This article is a science-first exploration of five peptide families with the most robust evidence bases for endurance-relevant physiology: collagen peptides (the only class with extensive human RCT data), MOTS-c (the mitochondrial exercise mimetic), BPC-157 (the gastric cytoprotective peptide), TB-500 / Thymosin Beta-4 (the actin-regulating wound healer), and GHK-Cu (the copper-binding tissue remodeler). Along the way, we will examine how companies like Peptilogics, Menten AI, and Google DeepMind are using generative AI to design novel peptide sequences, and why tools like the Cardiorespiratory Digital Twin™ and the Digital Twin for Predictive Performance™ may be the missing link between laboratory peptide science and individualized athlete protocols.
The five peptide families most relevant to endurance athletes span a wide evidence spectrum. Collagen peptides are the only class with extensive human randomized controlled trial data and unrestricted WADA status. MOTS-c, BPC-157, and TB-500 remain prohibited under WADA's S0 Unapproved Substances category. GHK-Cu is currently not listed.
1. Collagen Peptides: The Only Peptide Class With Deep Human Trial Data
If peptide science were a courtroom, collagen peptides would be the prosecution's star witness. Daniel König, professor at the University of Vienna's Centre for Sports Science, has been at the center of this work, establishing that bioactive peptides derived from collagen hydrolysis can meaningfully influence connective-tissue remodeling, body composition, and recovery from exercise-induced muscle damage.
The Evidence: From Lab Bench to Running Track
A 2024 systematic review and meta-analysis published in Sports Medicine examined 19 RCTs investigating collagen peptide (CP) supplementation combined with physical training. The meta-analysis found that prolonged CP supplementation alongside resistance or concurrent training produced statistically significant improvements in fat-free mass, maximal strength, and tendon morphological adaptations. Most trials administered 15 grams of specific collagen peptides (SCP) daily, though emerging 2024 data suggests 30 grams may further enhance collagen synthesis in resistance-trained subjects.
For endurance athletes specifically, Centner, Jerger, and König published a randomized controlled trial in Nutrients (2023) examining recreationally active men who combined 12 weeks of concurrent training—60 minutes of moderate-intensity running plus 15 minutes of dynamic resistance work—with daily SCP supplementation. The treatment group demonstrated significant improvements in one-hour time-trial performance on a running track, along with favorable shifts at the aerobic and anaerobic thresholds. These were moderately trained runners showing measurable endurance gains that exceeded what concurrent training alone produced.
Recovery data is equally compelling. In a 2023 RCT at the University of Vienna, 55 male participants consuming 15 grams of SCP daily while performing concurrent training for 12 weeks showed significantly improved recovery markers—including maximal voluntary contraction, rate of force development, and countermovement jump height—following 150 drop jumps. A companion study from the same group demonstrated that the SCP group had significantly lower blood levels of myoglobin, creatine kinase, and lactate dehydrogenase after a second bout of muscle-damaging exercise, indicating reduced acute muscle stress and improved musculotendinous structural integrity.
Twelve weeks of daily 15-gram collagen peptide supplementation combined with concurrent training significantly reduced exercise-induced muscle-damage markers and improved one-hour running time-trial performance in recreationally active men. Data adapted from König's research group at the University of Vienna.
The Mechanism: Extracellular Matrix as Performance Infrastructure
Collagen constitutes approximately 30 percent of total protein mass in the human body and 65 to 80 percent of tendon dry weight. The working hypothesis is that SCP supplementation increases the availability of amino acid precursors—glycine, proline, and hydroxyproline—that support training-induced adaptation of collagenous tissues. The extracellular matrix (ECM) is not merely scaffolding; it is the force-transmission highway through which myofibrillar proteins generate movement. Remodeling the ECM through targeted nutrition could enhance explosive-force generation and reduce susceptibility to the overuse injuries that plague distance athletes.
For the endurance Coach / Practitioner building periodized nutrition plans, collagen peptides represent the lowest-risk, highest-evidence peptide option. They are food-grade supplements, not prohibited by WADA, and can be timed around training sessions. When layered with wearable-derived recovery data inside a Cardiorespiratory Digital Twin™, the dosing of collagen supplementation can be personalized to each Athlete / Patient's training load and recovery trajectory.
2. MOTS-c: The Mitochondrial Exercise Mimetic That Rewrites Endurance Biology
If collagen peptides are the established veteran, MOTS-c is the electrifying rookie. Discovered in 2015 by Changhan David Lee and Pinchas Cohen at USC, MOTS-c is encoded in the 12S rRNA region of the mitochondrial genome—making it one of a small but growing family of mitochondrial-derived peptides (MDPs) that challenge the longstanding view of mitochondria as passive energy factories.
"This discovery sheds new light on mitochondria and positions them as active regulators of metabolism." — Changhan David Lee, Assistant Professor of Gerontology, USC Leonard Davis School
The Exercise Connection
Human studies show that circulating endogenous MOTS-c levels increase approximately 1.6-fold during exercise and remain elevated about 1.5-fold post-exercise before returning to baseline after roughly four hours. This temporal profile suggests MOTS-c functions as an exercise-induced mitokine—a signaling molecule released by mitochondria in working muscles that communicates metabolic status to the rest of the body.
In the 2021 Nature Communications study, Reynolds, Lai, and Lee demonstrated that MOTS-c treatment significantly enhanced physical performance in young, middle-aged, and old mice. Most strikingly, late-life-initiated intermittent treatment—started at 23.5 months, the murine equivalent of late human life—increased physical capacity and healthspan. Lee noted: "As we age, that communication network seems to break down, but our study suggests you can restore that network or rejuvenate an older mouse so it is as fit as a younger one."
A December 2024 study from Shanghai University of Sport tested serum MOTS-c levels in marathon runners versus sedentary subjects, finding a close association between circulating MOTS-c and aerobic exercise capacity. The authors proposed that MOTS-c levels could serve as a predictive indicator for aerobic capacity, body-fat status, and endurance training load. This raises an intriguing possibility for endurance practitioners: MOTS-c as a blood-based biomarker for mitochondrial fitness, complementing VO₂max and lactate threshold in a multi-modal assessment framework.
MOTS-c is a mitochondrial-derived peptide that translocates to the nucleus under metabolic stress, activating AMPK-dependent pathways that enhance glucose metabolism, fatty acid oxidation, and mitochondrial biogenesis—the core metabolic adaptations of endurance training.
The AMPK Connection and Metabolic Flexibility
MOTS-c operates primarily through the AMPK signaling pathway—the same master metabolic switch activated by exercise, caloric restriction, and metformin. Under metabolic stress, MOTS-c translocates from the cytoplasm to the nucleus, directly regulating gene expression related to stress adaptation. This retrograde signaling mechanism—mitochondria communicating upstream to the nucleus—is what Bérénice Benayoun, a faculty member at USC Leonard Davis and co-author on the Reynolds et al. study, has been investigating in the context of age-related decline.
For endurance athletes, the implications are substantial. Metabolic flexibility—the ability to switch efficiently between carbohydrate and fat oxidation—is arguably the most important metabolic attribute for ultra-distance performance. MOTS-c's demonstrated ability to enhance both glucose utilization and fatty acid oxidation positions it as a potential modulator of this flexibility. Troy L. Merry and Cameron Mitchell at the University of Auckland, collaborators on the Reynolds et al. study, have been exploring how MOTS-c expression varies with different training modalities and how ethnic-specific mitochondrial DNA variants may influence individual responses.
MOTS-c is not approved for human therapeutic use and is prohibited under the WADA S0 Unapproved Substances category. No human clinical trials of exogenous MOTS-c for athletic performance have been published. But it is precisely the kind of science that Calico Life Sciences—Alphabet's longevity research subsidiary—and Insilico Medicine are watching closely, because it suggests the mitochondrial genome harbors a pharmacopoeia of untapped bioactive molecules.
Circulating MOTS-c levels rise approximately 1.6-fold during exercise and 1.5-fold post-exercise before returning to baseline. Marathon runners show higher resting levels than sedentary controls, suggesting MOTS-c may serve as a blood-based biomarker for mitochondrial fitness and endurance capacity.
3. BPC-157: The Croatian Gastric Peptide That Became a Gray-Market Phenomenon
No peptide generates more debate than Body Protection Compound 157. A synthetic pentadecapeptide (Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val), BPC-157 was first isolated from human gastric juice by Predrag Sikiric and Sven Seiwerth at the University of Zagreb School of Medicine in the early 1990s. Sikiric's laboratory has since published more than 200 studies on the compound—an extraordinary body of work that has attracted both admiration for its breadth and criticism for originating almost entirely from a single research group.
What the Preclinical Data Shows
A 2025 systematic review in HSS Journal by Vasireddi, Hahamyan, Salata, and colleagues at Case Western Reserve University identified 36 studies published between 1993 and 2024. In animal models, BPC-157 improved functional, structural, and biomechanical outcomes in muscle, tendon, ligament, and bony injuries. The peptide appears to exert effects through upregulation of VEGF (angiogenesis), activation of FAK/paxillin pathways (cell adhesion and proliferation), stimulation of nitric oxide synthesis (cytoprotection), and increased growth hormone receptor gene expression.
The musculoskeletal findings are notable: in Achilles tendon transection models, BPC-157 accelerated granulation tissue formation, improved collagen organization, and promoted endothelial protection. In muscle-to-tendon junction injury models, therapy restored tissue continuity within six weeks without ectopic bone formation. The peptide is also remarkably stable in gastric juice—intact for over 24 hours—distinguishing it from most growth factors that are rapidly degraded in the GI tract.
BPC-157's research trajectory spans three decades and more than 200 publications—nearly all preclinical and originating from the University of Zagreb. Despite WADA prohibition since 2022 and no FDA approval, gray-market availability and social-media virality have made it one of the most discussed peptides in endurance and regenerative medicine.
The Human Evidence Gap
Despite the voluminous preclinical literature, published human trial data is nearly nonexistent. The sole human study identified by the 2025 systematic review was a retrospective case series of 12 patients who received intra-articular BPC-157 injections for knee pain; 7 of 12 reported subjective improvement at six-month follow-up. A separate 2025 pilot evaluated intravenous infusion at 10 mg and 20 mg in two adults with no adverse effects on organ biomarkers—but two subjects do not constitute a trial.
BPC-157 is prohibited under WADA's S0 category. The FDA has confirmed there is no legal basis for compounding pharmacies to use it. As USADA stated: the compound is not approved for human clinical use by any global regulatory authority. The NFL, UFC, and NCAA have all banned it since 2022.
As the Arthroscopy editorial by Dwyer et al. concluded in 2024: "We do not recommend the use of BPC-157 for sports performance and recovery because there are no randomized controlled trials in human subjects... However, with further research, therapeutic peptides may offer a synthetic alternative or adjunct to regenerative medicine."
4. TB-500 / Thymosin Beta-4: The Actin Regulator With a Wound-Healing Legacy
Thymosin Beta-4 is a 43-amino-acid peptide and one of the most abundant intracellular proteins in mammalian cells, playing a critical role in regulating actin polymerization—the molecular machinery driving cell movement, division, and tissue repair. TB-500, its synthetic fragment, gained scientific credibility when Malinda et al. demonstrated in 1999 that topical or intraperitoneal administration increased re-epithelialization by 42 percent over saline controls at day 4 and by 61 percent at day 7 in a rat wound model. Allan Goldstein at George Washington University, who pioneered the isolation of thymosins, described thymosin beta-4 as a "multi-functional regenerative peptide" with applications from cardiac repair to corneal healing.
Relevance to Endurance Athletes
The endurance rationale for TB-500 centers on three effects: angiogenesis promotion (new blood vessel formation improving tissue perfusion), inflammation modulation (calming excessive responses that delay recovery), and anti-fibrotic activity (reducing scar tissue that causes stiffness and re-injury risk). In preclinical models, accelerated healing of connective tissue and improved muscle recovery after endurance exercise have been reported.
A critical caveat: thymosin beta-4 is upregulated in many metastatic cancers, facilitating tumor-cell migration to distant sites. This theoretical cancer-promotion risk is a serious safety consideration that distinguishes TB-500 from other compounds in this article. Like BPC-157, it is WADA-prohibited and lacks any published human athletic-performance trials. The responsible path for athletes and their practitioners is to monitor the clinical-trial landscape while focusing on approaches with established safety profiles—PRP, evidence-based rehabilitation, and collagen peptide supplementation.
TB-500 promotes tissue repair through actin regulation, angiogenesis, and anti-fibrotic activity. However, thymosin beta-4's known upregulation in metastatic cancers represents a significant theoretical safety concern. No human athletic-performance trials have been published.
5. GHK-Cu: The Copper Peptide That Rewires Gene Expression for Tissue Repair
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a naturally occurring tripeptide first isolated in 1973 by biochemist Loren Pickart, who observed that plasma from younger individuals stimulated regenerative processes more effectively than plasma from older adults. Tracing this activity to a small copper-binding peptide, Pickart launched over five decades of research into a molecule with an unusually broad biological profile: it stimulates blood vessel and nerve outgrowth, increases collagen, elastin, and glycosaminoglycan synthesis, and supports dermal fibroblast function.
The Gene-Expression Story
What distinguishes GHK-Cu is the scale of its gene-regulatory effects. Analyses using the Broad Institute's Connectivity Map suggest GHK may regulate more than 4,000 human genes, resetting aging-associated patterns toward profiles characteristic of younger tissue. In human plasma, GHK concentrations decline from approximately 200 ng/mL at age 20 to roughly 80 ng/mL by age 60—a decline coinciding with the noticeable decrease in regenerative capacity that every aging athlete experiences firsthand.
Endurance Implications
The GHK-Cu story intersects with the collagen-peptide story at the level of connective-tissue maintenance. Repetitive-impact sports place enormous cumulative stress on tendons, ligaments, and the ECM. Age-related decline in endogenous GHK may contribute to progressive loss of tissue-repair capacity in masters-age athletes. While GHK-Cu has been studied primarily in dermatological contexts, its upregulation of collagen types I, III, and IV, along with fibronectin and proteoglycans, suggests potential musculotendinous relevance that awaits systematic investigation in athletic populations.
Notably, GHK-Cu is not on the 2025 WADA Prohibited List—one of the few compounds in this article that retains that distinction. It is widely available in topical formulations with an excellent safety profile established over decades of cosmetic use. Subcutaneous administration remains experimental, but for athletes, GHK-Cu represents an accessible and legally unrestricted compound for further exploration, particularly in the context of skin health and superficial tissue repair during high-volume training blocks.
Plasma GHK-Cu concentrations decline by more than 60 percent between ages 20 and 60, a trajectory that tracks closely with the loss of regenerative capacity. For aging endurance athletes, this decline may contribute to the increased vulnerability to overuse injuries that characterizes masters-level competition.
6. The AI Revolution in Peptide Design: From AlphaFold to Athlete-Specific Molecules
The five peptides above were all discovered through traditional biochemical methods. The next generation of endurance-relevant peptides may emerge from AI-driven computational design.
Google DeepMind's AlphaFold solved the protein-structure prediction problem with a computational accuracy that stunned structural biology and spawned a new wave of peptide-design companies. Peptilogics, based in Pittsburgh, uses generative AI models and reinforcement learning to explore enormous peptide chemical spaces, generating sequences optimized for anti-inflammatory and tissue-repair targets directly relevant to athletic recovery. Menten AI in Toronto applies machine-learning-guided protein design to create peptides with desired pharmacological properties, leveraging structure-based modeling for binding affinity and metabolic stability. Aizen Therapeutics uses expanded amino-acid chemistries to design peptides beyond the 20 standard amino acids, potentially creating molecules with enhanced stability and bioactivity.
ProteinQure, an early entrant in AI-native peptide therapeutics, combines physics-based molecular dynamics with data-driven optimization. XtalPi's PepiX platform integrates quantum chemistry with AI to predict peptide conformations and binding properties, compressing design-to-synthesis timelines from years to months. Stanford University's bioengineering and peptide therapeutics groups have been exploring how computational peptide design can integrate with wearable-derived physiological data to create personalized recovery molecules. The Scripps Research Institute has been investigating similar approaches, using high-throughput screening with AI-guided optimization to identify peptides targeting inflammatory pathways activated during prolonged endurance exercise.
For major pharmaceutical companies—Novo Nordisk, AstraZeneca, and Sanofi among them—peptide therapeutics represent a growing priority. The market has been expanding at roughly 9 percent annually since 2015. Much of that growth is driven by GLP-1 receptor agonists like semaglutide, but the next wave may include performance- and recovery-oriented peptides designed for active and aging populations.
The AI peptide discovery pipeline is compressing the journey from target identification to candidate molecule from decades to months. Companies like Peptilogics, Menten AI, and ProteinQure are applying generative models and reinforcement learning to design novel peptides—some with potential endurance-recovery applications that traditional biochemistry would have taken years to identify.
7. Personalized Peptide Protocols and the Digital Twin for Predictive Performance™
The fundamental challenge with peptide science—as with all interventions in sports medicine—is individual variability. Two athletes with identical VO₂max values, training volumes, and diets may respond differently to collagen peptide supplementation, because their connective-tissue genetics, inflammatory profiles, and gut-absorption kinetics differ.
A Cardiorespiratory Digital Twin™ can ingest wearable data (heart-rate variability, sleep architecture, training load), biomarker data (inflammatory markers, MOTS-c levels, collagen-turnover markers), and genomic data (connective-tissue gene variants, mitochondrial DNA haplogroup) to create a continuously updated model of an individual athlete's physiological state. Within this framework, peptide supplementation strategies can be simulated, optimized, and personalized—moving beyond "15 grams of collagen for everybody" toward dosing calibrated to each athlete's recovery dynamics.
Companies in the longevity-tech ecosystem are approaching this from complementary angles. Fountain Life, co-founded by Peter Diamandis, offers comprehensive biomarker panels that can track collagen-turnover markers longitudinally. InsideTracker provides blood-based performance optimization that could incorporate peptide-relevant biomarkers. Function Health runs extensive panels creating longitudinal profiles. Deep Longevity uses AI to analyze biological aging clocks. TruDiagnostic provides epigenetic-age testing. Viome profiles the gut microbiome—potentially relevant to oral peptide absorption kinetics. When these data streams converge inside a digital twin, the result is the level of personalization that transforms peptide science from population-level generalizations into individual-level prescriptions—the same philosophy driving Andrew Huberman's evidence-based protocols, Peter Attia's emphasis on precision medicine, and the monitoring frameworks used by Stanford Athletics and NASA.
A Cardiorespiratory Digital Twin™ integrates wearable, biomarker, genomic, and microbiome data streams to personalize peptide supplementation protocols for individual endurance athletes—moving beyond generic dosing toward precision interventions calibrated to each athlete's recovery dynamics and training load.
8. The Regulatory Reality: What Every Endurance Athlete Must Know
The current regulatory state of play, as of early 2026:
Collagen peptides are classified as food supplements in most jurisdictions, are not prohibited by WADA, and carry minimal regulatory risk for competitive athletes.
MOTS-c is prohibited under WADA's S0 Unapproved Substances category. Not approved for human therapeutic use in any country. No basis for a Therapeutic Use Exemption.
BPC-157 is prohibited under WADA's S0 category. The FDA has confirmed no legal basis for selling it as a drug, food, supplement, or compounded medication.
TB-500 / Thymosin Beta-4 is prohibited under WADA's Peptide Hormones, Growth Factors, and Related Substances category. FDA Category 2 bulk drug substance; not approved for human therapeutic use.
GHK-Cu is not on the 2025 WADA Prohibited List. Available in cosmetic formulations. Athletes should verify annually as the list is updated.
The regulatory picture underscores why evidence-based endurance coaching and proactive longevity planning matter. An endurance Coach / Practitioner who stays current on WADA regulations, FDA guidance, and the clinical-trial pipeline can help athletes navigate this landscape without jeopardizing careers, health, or competitive eligibility.
9. Conclusion: The Peptide Future Is Closer Than You Think—If You Plan for It
The five peptide families explored here represent a spectrum from established to experimental: collagen peptides sit at the evidence-rich, regulatory-safe end; MOTS-c, BPC-157, TB-500, and GHK-Cu occupy different positions along a continuum of preclinical promise and clinical-evidence deficit. What unites them is the underlying biology: short chains of amino acids functioning as precision signaling molecules, communicating repair instructions, metabolic adjustments, and adaptive responses at the cellular level.
For the endurance athlete—whether a 28-year-old marathoner chasing a qualifying time or a 62-year-old executive training for their first Ironman as a longevity strategy—the practical takeaway is threefold. First, collagen peptide supplementation has sufficient evidence to warrant inclusion in well-designed training nutrition, ideally timed around sessions and calibrated to load. Second, the emerging science on mitochondrial-derived peptides like MOTS-c suggests that our understanding of endurance biology is about to undergo a fundamental expansion—one likely to yield new biomarkers, training-response models, and eventually therapeutic options. Third, the gray-market peptide economy poses real risks: regulatory, health-related, and career-ending for competitive athletes. The responsible path is to monitor clinical trials, support rigorous research, and avoid unregulated compounds while the science matures.
"The future of athletic longevity is not a single intervention—it is a system. A system integrating wearable data, blood biomarkers, genomic insights, and AI-powered modeling into a personalized, continuously updated plan. Peptide science is one input into that system. The Digital Twin for Predictive Performance™ is the computational engine that personalizes it for the Athlete / Patient. And the endurance Coach / Practitioner who understands both the science and the system will be the one who helps their athletes not just perform, but recover, succeed and endure." — Tony Medrano, CEO, LongevityPlan.AI
Planning for your longevity is not a luxury—it is a strategy. And in the peptide era, the athletes who thrive will be the ones who bring data, science, and personalization to every decision they make.
The future of endurance performance is not a single peptide—it is a system integrating molecular science, real-time wearable data, blood biomarkers, and AI-powered Digital Twins into personalized protocols that help athletes compete longer and recover smarter.
Endnotes
- Lee, C., Zeng, J., Drew, B.G., et al. (2015). "The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance." Cell Metabolism, 21(3), 443–454.
- Reynolds, J.C., Lai, R.W., Woodhead, J.S.T., et al. (2021). "MOTS-c is an exercise-induced mitochondrial-encoded regulator of age-dependent physical decline and muscle homeostasis." Nature Communications, 12, 470.
- Sikiric, P., Rucman, R., Turkovic, B., et al. (2018). "Novel cytoprotective mediator, stable gastric pentadecapeptide BPC 157." Current Pharmaceutical Design, 24(18), 1990–2001.
- Dwyer, M.T.D., et al. (2024). "Injectable Therapeutic Peptides—An Adjunct to Regenerative Medicine and Sports Performance?" Arthroscopy, 41(2), 150–152.
- Vasireddi, N., Hahamyan, H., Salata, M.J., et al. (2025). "Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review." HSS Journal, 21, 485–495.
- König, D., Kohl, J., Jerger, S., & Centner, C. (2021). "Potential Relevance of Bioactive Peptides in Sports Nutrition." Nutrients, 13(11), 3997.
- Centner, C., et al. (2024). "Impact of Collagen Peptide Supplementation in Combination with Long-Term Physical Training." Sports Medicine, 54, 2669–2690.
- Centner, C., Jerger, S., et al. (2023). "Effects of Specific Bioactive Collagen Peptides in Combination with Concurrent Training on Running Performance." Nutrients, 15(20), 4300.
- Jerger, S., Centner, C., et al. (2023). "Influence of specific collagen peptides and 12-week concurrent training on recovery-related biomechanical characteristics." Frontiers in Nutrition, 10, 1266056.
- Bischof, K., Stafilidis, S., et al. (2024). "Reduction in systemic muscle stress markers after exercise-induced muscle damage." Frontiers in Nutrition, 11, 1384112.
- Khatri, M., Naughton, R.J., Clifford, T., et al. (2021). "The effects of collagen peptide supplementation on body composition, collagen synthesis, and recovery." Amino Acids, 53, 1493–1506.
- USC Pressroom (2015). "Newly Discovered Hormone Mimics the Effects of Exercise."
- Frontiers in Endocrinology (2023). "MOTS-c: A promising mitochondrial-derived peptide for therapeutic exploitation." 14, 1120533.
- ScienceDirect (2024). "Endurance training enhances skeletal muscle mitochondrial respiration by promoting MOTS-c secretion." Shanghai University of Sport.
- Merry, T.L., et al. (2020). "Mitochondrial-derived peptides in energy metabolism." Free Radical Biology and Medicine, 100, 182–187.
- "Emerging Anabolic and Regenerative Peptides in Athletic Body Re-composition and Bodybuilding." Genesis Publishing (2025).
- USADA (2025). "BPC-157: Experimental Peptide Creates Risk for Athletes."
- Malinda, K.M., Sidhu, G.S., Mani, H., et al. (1999). "Thymosin beta4 accelerates wound healing." Journal of Investigative Dermatology, 113(3), 364–368.
- Goldstein, A.L., Hannappel, E., Sosne, G., & Kleinman, H.K. (2012). "Thymosin β4: a multi-functional regenerative peptide." Expert Opinion on Biological Therapy, 12(1), 37–51.
- Preprints.org (2025). "Safety and Efficacy of Approved and Unapproved Peptide Therapies for Musculoskeletal Injuries and Athletic Performance."
- Pickart, L. (1973). "A Tripeptide from Human Serum Which Enhances the Growth of Neoplastic Hepatocytes." Nature New Biology, 243(124), 85–87.
- Pickart, L. & Margolina, A. (2018). "Regenerative and Protective Actions of the GHK-Cu Peptide." International Journal of Molecular Sciences, 19(7), 1987.
- Pickart, L., Vasquez-Soltero, J.M., & Margolina, A. (2015). "GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration." BioMed Research International, 2015, 648108.
- López-Martínez, M.I., Miguel, M., & Garcés-Rimón, M. (2022). "Protein and Sport: Alternative Sources and Strategies for Bioactive and Sustainable Sports Nutrition." Frontiers in Nutrition, 9, 926043.
- Penggalih, M.H.S.T., et al. (2023). "Marine-derived protein: peptide bioresources for the development of nutraceuticals for improved athletic performance." Frontiers in Sports and Active Living, 5, 1281397.
About the Author
Tony Medrano is CEO and co-founder of LongevityPlan.AI, a platform that integrates performance and health data from athletes and leverages proprietary Cardiorespiratory Digital Twin™ technology, wearable data, and biomarker data to deliver personalized performance optimization and longevity recommendations to athletes, coaches, organizations, businesses, government, and the military. In addition to being a 3x technology / AI company CEO with 2 successful exits, Tony has also finished 3 Full Ironman Triathlons (140.6 mi) since 2019. He has degrees from Harvard University, Columbia University, and a JD/MBA from Stanford University.