Clinical Research
·5 min read
How Digital Twins Could Redefine Clinical Trials
Exploring the Shift from Reactive Trials to Predictive Modeling
By Tony Medrano & Taylor Barkdoll, LongevityPlan.AI
Exploring the Shift from Reactive Trials to Predictive Modeling
By Tony Medrano, CEO & Co-Founder, and Taylor Barkdoll, Biology Market Development, LongevityPlan.AI
1. The $2.6 Billion Coin Flip: Why Clinical Trials Are Broken
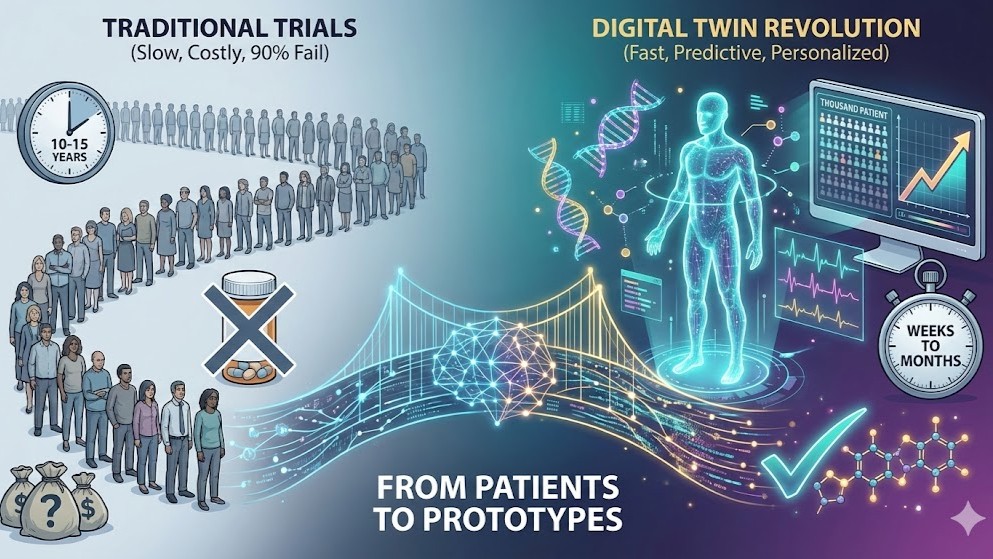
Here is a number that should keep every pharmaceutical executive, health-conscious investor, and aspiring centenarian awake at night: 90 percent of drug candidates that enter clinical trials never reach patients. The average cost of shepherding a single molecule from laboratory bench to pharmacy shelf now exceeds $2.6 billion, a figure that has tripled in inflation-adjusted dollars since the early 2000s. The timeline stretches 10 to 15 years. And the human toll—hundreds of thousands of volunteers enrolled in trials that ultimately fail—is incalculable.
The arithmetic gets worse the deeper you look. A typical Phase III oncology trial requires 500 to 3,000 patients. Recruitment alone can take two to four years. The cost per enrolled patient averages $42,000, but in complex therapeutic areas like respiratory disease, total per-study costs can exceed $115 million. And after all of that investment, approximately 50 percent of Phase III trials still fail.
Analyses of clinical trial data from 2010 to 2017 identified four primary reasons for this carnage: lack of clinical efficacy (40–50 percent of failures), unmanageable toxicity (30 percent), poor drug-like properties (10–15 percent), and inadequate strategic planning (10 percent). Notice that not one of these failure modes is insurmountable with better predictive modeling.
The Alzheimer's disease space illustrates the stakes with particular clarity. Between 2002 and 2012, the failure rate for Alzheimer's drugs was 99.6 percent. Every failed trial represents not only billions in sunk costs but years of lost hope for patients and families. If digital twins can even modestly improve the probability of success—say, from 10 percent to 15 percent—the downstream economic and human impact is staggering.
Enter the digital twin.
2. Digital Twins 101: From Rocket Science to Your Bloodstream
Originally engineered by NASA to monitor spacecraft in orbit—creating virtual replicas of physical systems that could be stress-tested without risking hardware—digital twin technology is now migrating from aerospace hangars into hospital wards, pharmaceutical boardrooms, and, increasingly, into your personal health planning toolkit.
A digital twin in medicine is a computational model of all or part of a patient, continuously calibrated with real-world data, that can simulate responses to interventions before those interventions are ever administered. Think of it as a flight simulator for your biology.
The term "digital twin" was formalized by Michael Grieves at the University of Michigan in 2002, though NASA had been using the concept since the Apollo program. In its industrial incarnation, a digital twin has two defining features: it is built on a mechanistic model of the physical system, and it is dynamically calibrated to that system through a continuous, bidirectional flow of data.
Medicine adopted the paradigm more recently but with explosive momentum. A PubMed search for "digital twin" now returns over 1,400 citations, with an exponential increase since 2020.
Three Layers of Medical Digital Twins
The 2025 Lancet Digital Health review proposes a useful framework. Medical digital twins can operate at three scales:
Molecular-level twins model cellular processes: gene expression, protein folding, metabolic flux. These are the engines behind AI-driven drug discovery at companies like Insilico Medicine and Roche.
Organ-level twins replicate the physics of specific organs—the beating heart, the ventilating lung, the filtering kidney. Dassault Systèmes' Living Heart Project, for instance, creates patient-specific cardiac simulations used by the FDA to evaluate medical devices.
Whole-patient twins integrate multi-modal data—genomics, wearables, imaging, electronic health records—into a holistic avatar. This is the frontier where clinical trials and personal longevity planning converge, and where the concept of a Cardiorespiratory Digital Twin™ begins to show its transformative potential.
3. Unlearn.AI and the Art of the Virtual Placebo
Perhaps no company has done more to operationalize digital twins in clinical trials than San Francisco-based Unlearn.AI. Founded by Charles K. Fisher, a physicist turned machine-learning scientist, Unlearn has built a platform called TwinRCTs that creates individual-level digital twins of clinical trial participants—specifically, AI-generated predictions of how each participant would have progressed if they had received a placebo.
The regulatory milestones are significant. In September 2022, the European Medicines Agency (EMA) formally qualified PROCOVA for use as the primary analysis methodology in Phase 2 and Phase 3 clinical trials with continuous outcomes. The U.S. Food and Drug Administration (FDA) subsequently confirmed that PROCOVA aligns with its current guidance on covariate adjustment. These are not pilot endorsements. They are regulatory green lights for real trials with real patients.
"This variance reduction [with digital twins] could have had a significant impact on the number of subjects we needed…and still preserved the same power…we would have had faster enrollment, encouraged greater patient participation. And ultimately, that would have been cost saving and time saving." — Ole Graff, Executive Medical Director of Neuroscience, AbbVie
A concrete demonstration of the technology's power emerged in late 2025, when AbbVie and Unlearn published results showing that digital twins, applied retroactively to the 453-patient AWARE trial, reduced treatment-effect variance and offered the potential for meaningful sample-size reductions—without inflating Type I error rates.
What This Means for Longevity Planning
This article examines how digital twins are poised to redefine clinical trials—and, by extension, the entire trajectory of personalized and preventive medicine. The convergence of regulatory approval, demonstrated clinical utility, and advancing AI capabilities suggests we are at an inflection point.
For the longevity-minded, this distinction matters enormously. A static snapshot of your blood panel is a photograph. A digital twin is a motion picture, complete with the ability to fast-forward and ask: What happens to my cardiovascular system if I add Zone 2 training three days per week? What if I start metformin at age 52 versus 58?
Read the full article on LinkedIn for the complete analysis including in silico trials, regulatory frameworks, and future implications.