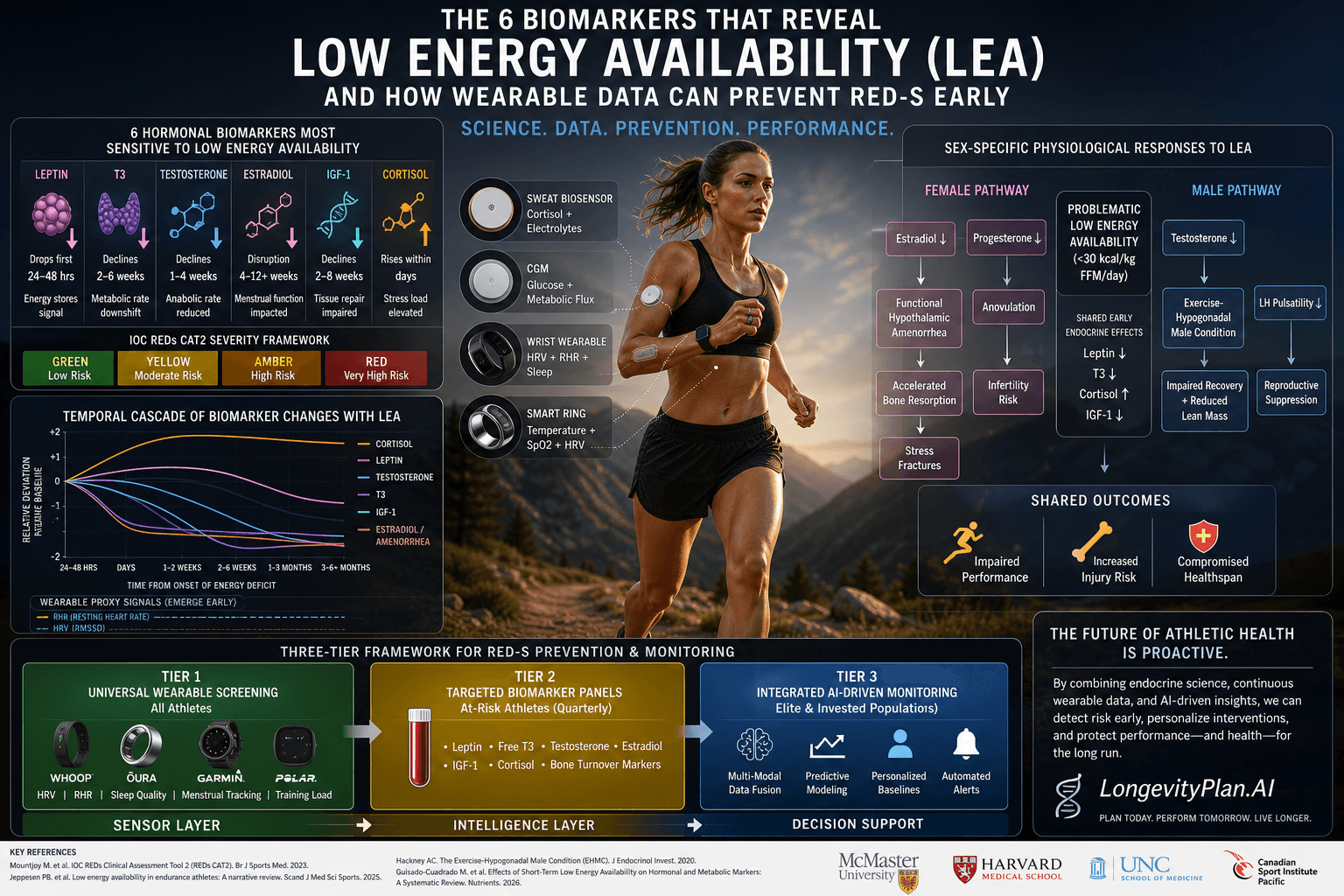
Health Science
·3 min read
Molecular Mystery of Chronic Fatigue
How Disrupted Cellular Energy Production, WASF3 Protein Dysfunction, and AI-Powered Cardiorespiratory Digital Twins™ Are Rewriting the Future of Fatigue Medicine
By Tony Medrano & Sydney Wiredu, LongevityPlan.AI
How Disrupted Cellular Energy Production, WASF3 Protein Dysfunction, and AI-Powered Cardiorespiratory Digital Twins™ Are Rewriting the Future of Fatigue Medicine
By Tony Medrano & Sydney Wiredu, LongevityPlan.AI
The Invisible Epidemic
Chronic fatigue affects millions worldwide, yet its molecular origins have remained frustratingly elusive—until now. Groundbreaking research into WASF3 protein dysfunction and disrupted mitochondrial energy production is finally illuminating the cellular mechanisms behind this debilitating condition.
For decades, chronic fatigue syndrome (CFS/ME) was dismissed as psychosomatic, leaving patients without answers or effective treatments. But the convergence of molecular biology and AI-powered health monitoring is changing everything.
The WASF3 Connection
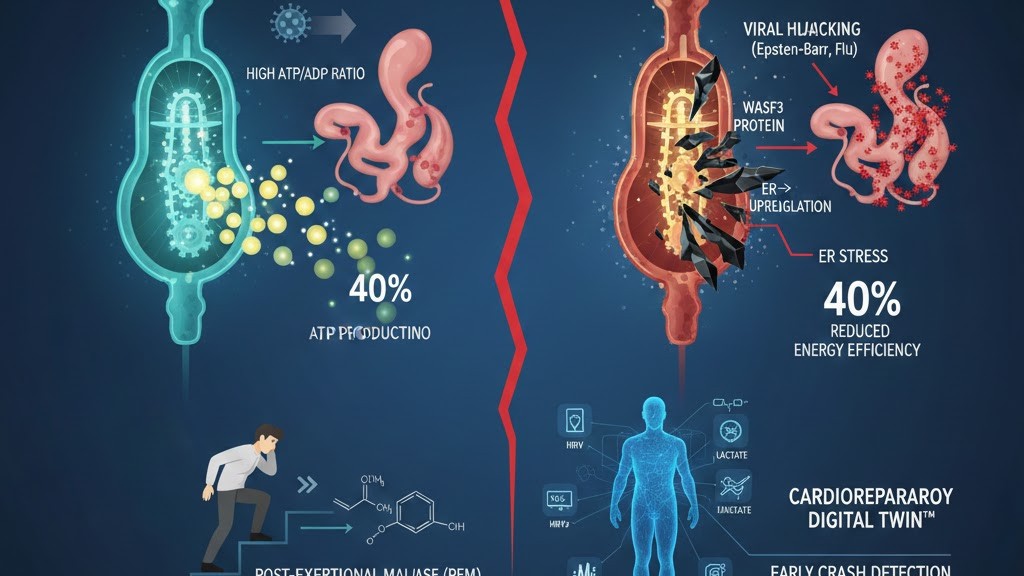
Recent NIH-funded research has identified WASF3, a protein involved in mitochondrial function, as a key player in chronic fatigue. When WASF3 levels become dysregulated, mitochondrial energy production falters—leaving cells unable to meet the body's energy demands. The research, published in Proceedings of the National Academy of Sciences (PNAS), demonstrated that overexpression of WASF3 disrupts Complex III of the electron transport chain, reducing ATP output.
This isn't just about feeling tired. It's about fundamental cellular dysfunction that affects every system in the body: cardiovascular output, cognitive function, immune response, and metabolic efficiency. Dr. Avindra Nath, Clinical Director at the National Institute of Neurological Disorders and Stroke (NINDS), led the NIH intramural study that identified WASF3's role.
Digital Twins Meet Molecular Medicine
This is where AI-powered Cardiorespiratory Digital Twins™ enter the picture. By continuously monitoring heart rate variability, respiratory patterns, oxygen utilization, and recovery dynamics, these virtual replicas can detect the subtle physiological signatures of mitochondrial dysfunction before traditional tests can.
Imagine having a system that can:
- Track your cellular energy production efficiency in real-time
- Identify patterns that correlate with WASF3-related dysfunction
- Predict fatigue episodes before they occur
- Optimize training and recovery to work within your current mitochondrial capacity
Platforms like WHOOP, Oura, and Garmin already track proxies for mitochondrial function through HRV and respiratory rate. Dr. Peter Attia has extensively discussed how declining VO2 max—fundamentally a measure of mitochondrial oxygen utilization—serves as both a fatigue indicator and the strongest predictor of all-cause mortality.
The Future of Fatigue Medicine
The integration of molecular insights with AI-powered monitoring represents a paradigm shift in how we understand and treat fatigue disorders. Rather than treating symptoms, we can now target the root cause: disrupted cellular energy production.
Organizations like the Solve ME/CFS Initiative and the Open Medicine Foundation are funding research into biomarker identification. Stanford University's Dr. Ron Davis has pioneered nanotechnology-based blood tests that can distinguish ME/CFS patients from healthy controls—a potential diagnostic breakthrough. Dr. Robert Naviaux at UC San Diego has identified the cell danger response (CDR) as a unifying mechanism.
For athletes, executives, and health-conscious individuals, this means earlier detection of energy system dysfunction, personalized protocols based on your unique mitochondrial profile, optimized performance within your actual physiological capacity, and prevention of overtraining and burnout.
The molecular mystery of chronic fatigue is being solved—and AI Digital Twins are the key to translating that knowledge into actionable, personalized health optimization.